Effect of vitamin D3 on asthma treatment failures in adults with symptomatic asthma and lower vitamin D levels: the VIDA randomized clinical trial
- PMID: 24838406
- PMCID: PMC4217655
- DOI: 10.1001/jama.2014.5052
Effect of vitamin D3 on asthma treatment failures in adults with symptomatic asthma and lower vitamin D levels: the VIDA randomized clinical trial
Abstract
Importance: In asthma and other diseases, vitamin D insufficiency is associated with adverse outcomes. It is not known if supplementing inhaled corticosteroids with oral vitamin D3 improves outcomes in patients with asthma and vitamin D insufficiency.
Objective: To evaluate if vitamin D supplementation would improve the clinical efficacy of inhaled corticosteroids in patients with symptomatic asthma and lower vitamin D levels.
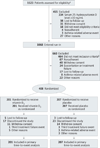
Design, setting, and participants: The VIDA (Vitamin D Add-on Therapy Enhances Corticosteroid Responsiveness in Asthma) randomized, double-blind, parallel, placebo-controlled trial studying adult patients with symptomatic asthma and a serum 25-hydroxyvitamin D level of less than 30 ng/mL was conducted across 9 academic US medical centers in the National Heart, Lung, and Blood Institute's AsthmaNet network, with enrollment starting in April 2011 and follow-up complete by January 2014. After a run-in period that included treatment with an inhaled corticosteroid, 408 patients were randomized.
Interventions: Oral vitamin D3 (100,000 IU once, then 4000 IU/d for 28 weeks; n = 201) or placebo (n = 207) was added to inhaled ciclesonide (320 µg/d). If asthma control was achieved after 12 weeks, ciclesonide was tapered to 160 µg/d for 8 weeks, then to 80 µg/d for 8 weeks if asthma control was maintained.
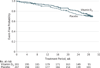
Main outcomes and measures: The primary outcome was time to first asthma treatment failure (a composite outcome of decline in lung function and increases in use of β-agonists, systemic corticosteroids, and health care).
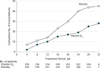
Results: Treatment with vitamin D3 did not alter the rate of first treatment failure during 28 weeks (28% [95% CI, 21%-34%] with vitamin D3 vs 29% [95% CI, 23%-35%] with placebo; adjusted hazard ratio, 0.9 [95% CI, 0.6-1.3]). Of 14 prespecified secondary outcomes, 9 were analyzed, including asthma exacerbation; of those 9, the only statistically significant outcome was a small difference in the overall dose of ciclesonide required to maintain asthma control (111.3 µg/d [95% CI, 102.2-120.4 µg/d] in the vitamin D3 group vs 126.2 µg/d [95% CI, 117.2-135.3 µg/d] in the placebo group; difference of 14.9 µg/d [95% CI, 2.1-27.7 µg/d]).
Conclusions and relevance: Vitamin D3 did not reduce the rate of first treatment failure or exacerbation in adults with persistent asthma and vitamin D insufficiency. These findings do not support a strategy of therapeutic vitamin D3 supplementation in patients with symptomatic asthma.
Trial registration: clinicaltrials.gov Identifier: NCT01248065.
Conflict of interest statement
Figures
References
-
- Nanzer AM, Chambers ES, Ryanna K, et al. Enhanced production of IL-17A in patients with severe asthma is inhibited by 1α,25-dihydroxyvitamin D3 in a glucocorticoid-independent fashion. J Allergy Clin Immunol. 2013;132(2):297–304. e3. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- UL1 TR000005/TR/NCATS NIH HHS/United States
- U10 HL098075/HL/NHLBI NIH HHS/United States
- HL098103/HL/NHLBI NIH HHS/United States
- U10 HL098096/HL/NHLBI NIH HHS/United States
- T32 HL007317/HL/NHLBI NIH HHS/United States
- U10 HL098102/HL/NHLBI NIH HHS/United States
- U10 HL098115/HL/NHLBI NIH HHS/United States
- HL098115/HL/NHLBI NIH HHS/United States
- UL1TR000439/TR/NCATS NIH HHS/United States
- UL1 TR001082/TR/NCATS NIH HHS/United States
- HL098112/HL/NHLBI NIH HHS/United States
- HL098107/HL/NHLBI NIH HHS/United States
- U10 HL098098/HL/NHLBI NIH HHS/United States
- UL1 TR000430/TR/NCATS NIH HHS/United States
- HL098075/HL/NHLBI NIH HHS/United States
- UL1TR000050/TR/NCATS NIH HHS/United States
- UL1TR000430/TR/NCATS NIH HHS/United States
- U10HL098096/HL/NHLBI NIH HHS/United States
- U10 HL098107/HL/NHLBI NIH HHS/United States
- UL1TR000454/TR/NCATS NIH HHS/United States
- UL1TR000150/TR/NCATS NIH HHS/United States
- UL1 TR000150/TR/NCATS NIH HHS/United States
- UL1TR001082/TR/NCATS NIH HHS/United States
- UL1 TR000427/TR/NCATS NIH HHS/United States
- U10 HL098177/HL/NHLBI NIH HHS/United States
- HL098098/HL/NHLBI NIH HHS/United States
- UL1TR000448/TR/NCATS NIH HHS/United States
- HL098090/HL/NHLBI NIH HHS/United States
- UL1 TR000050/TR/NCATS NIH HHS/United States
- HL098102/HL/NHLBI NIH HHS/United States
- UL1 TR000439/TR/NCATS NIH HHS/United States
- UL1 TR000454/TR/NCATS NIH HHS/United States
- U10 HL098103/HL/NHLBI NIH HHS/United States
- HL098177/HL/NHLBI NIH HHS/United States
- U10 HL074225/HL/NHLBI NIH HHS/United States
- UL1 TR000448/TR/NCATS NIH HHS/United States
- U10 HL098090/HL/NHLBI NIH HHS/United States
- U10 HL098112/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

