Insulin glargine in pediatric patients with type 1 diabetes in Japan
- PMID: 24840321
- PMCID: PMC4311442
- DOI: 10.1111/ped.12379
Insulin glargine in pediatric patients with type 1 diabetes in Japan
Abstract
Background: We evaluated the safety and effectiveness of insulin glargine in Japanese pediatric patients with type 1 diabetes in clinical settings based on post-marketing surveillance data.
Methods: Clinical data were collected from Japanese pediatric patients with type 1 diabetes for 24 weeks after initiation of glargine treatment. Baseline characteristics, hemoglobin A1c (HbA1c), fasting plasma glucose (FPG), previous/concomitant medication, height, bodyweight, and adverse events were analyzed.
Results: One-hundred and thirteen patients were enrolled from 20 medical institutions in Japan in 2003 and 2004. Of these patients, 73 were included in the safety analysis, and 70 of these patients were also included in the efficacy analysis. The 73 patients included 28 boys and 45 girls, with a mean age of 11.8 years at entry. Hypoglycemia occurred in three patients (three events) and was severe in two patients (two events); all patients recovered. In the efficacy evaluation, HbA1c at baseline and final assessment was 9.10% and 8.09% (P < 0.001) in all patients; 8.96% and 7.85% (P < 0.001) in patients aged 7-12 years (Group 1); and 9.28% and 8.37% (P = 0.010) in patients aged 13-15 years (Group 2). FPG significantly decreased in all patients and in Group 1. No significant changes were observed in body mass index or degree of obesity during the study.
Conclusions: Glargine therapy for Japanese pediatric patients with type 1 diabetes resulted in good glycemic control in terms of HbA1c and FPG as well as good safety in clinical settings. Glargine had little effect on the physical build of patients.
Keywords: degree of obesity; glycemic control; hypoglycemia; insulin glargine; pediatric patients with type 1 diabetes.
© 2014 The Authors. Pediatrics International published by Wiley Publishing Asia Pty Ltd on behalf of Japan Pediatric Society.
Figures
Similar articles
-
Long-term safety and effectiveness of biosimilar insulin glargine in Japanese patients with diabetes mellitus in routine clinical practice: results of a post-marketing safety study.Curr Med Res Opin. 2020 Jun;36(6):947-958. doi: 10.1080/03007995.2020.1754182. Epub 2020 Apr 23. Curr Med Res Opin. 2020. PMID: 32271092
-
Real-world data on the use of insulin glargine 300 U/mL in Japanese patients with type 1 diabetes: twelve-month results from a post-marketing surveillance study (X-STAR study).Expert Opin Pharmacother. 2021 Feb;22(2):249-256. doi: 10.1080/14656566.2020.1810665. Epub 2020 Aug 25. Expert Opin Pharmacother. 2021. PMID: 32840155
-
Effectiveness and safety of basal supported oral therapy with insulin glargine, in Japanese insulin-naive, type 2 diabetes patients, with or without microvascular complications: subanalysis of the observational, non-interventional, 24-week follow-up Add-on Lantus® to Oral Hypoglycemic Agents (ALOHA) study.J Diabetes Complications. 2015 Jan-Feb;29(1):127-33. doi: 10.1016/j.jdiacomp.2014.09.012. Epub 2014 Sep 28. J Diabetes Complications. 2015. PMID: 25449981
-
A randomized crossover study of the efficacy and safety of switching from insulin glargine to insulin degludec in children with type 1 diabetes.Endocr J. 2017 Feb 27;64(2):133-140. doi: 10.1507/endocrj.EJ16-0294. Epub 2016 Oct 14. Endocr J. 2017. PMID: 27746408 Clinical Trial.
-
[Use of insulin glargine in type 1 diabetes children with less than eight years old].Arq Bras Endocrinol Metabol. 2009 Aug;53(6):721-5. doi: 10.1590/s0004-27302009000600005. Arq Bras Endocrinol Metabol. 2009. PMID: 19893914 Clinical Trial. Portuguese.
Cited by
-
Impact of switching from neutral protamine hagedorn insulin (NPH) to glargine insulin on glycemic control and clinical outcomes in pediatric patients with type 1 diabetes.Explor Res Clin Soc Pharm. 2025 May 2;18:100612. doi: 10.1016/j.rcsop.2025.100612. eCollection 2025 Jun. Explor Res Clin Soc Pharm. 2025. PMID: 40491723 Free PMC article.
-
Switching From Glargine to Degludec: The Effect on Metabolic Control and Safety During 1-Year of Real Clinical Practice in Children and Adolescents With Type 1 Diabetes.Front Endocrinol (Lausanne). 2018 Aug 23;9:462. doi: 10.3389/fendo.2018.00462. eCollection 2018. Front Endocrinol (Lausanne). 2018. PMID: 30190702 Free PMC article.
References
-
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 1993;329:977–986. - PubMed
-
- Mortensen HB, Villumsen J, Volund A, Petersen KE, Nerup J. Relationship between insulin injection regimen and metabolic control in young Danish type 1 diabetic patients. The Danish Study Group of Diabetes in Childhood. Diabet. Med. 1992;9:834–839. - PubMed
-
- Mortensen HB, Hougaard P. Comparison of metabolic control in a cross-sectional study of 2,873 children and adolescents with IDDM from 18 countries. The Hvidore Study Group on Childhood Diabetes. Diabetes Care. 1997;20:714–720. - PubMed
-
- Mortensen HB, Robertson KJ, Aanstoot HJ, et al. Insulin management and metabolic control of type 1 diabetes mellitus in childhood and adolescence in 18 countries. Hvidore Study Group on Childhood Diabetes. Diabet. Med. 1998;15:752–759. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical


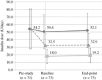
 …
…  ) and total insulin (
) and total insulin ( ) at pre-study, baseline, and end-point of the study.
) at pre-study, baseline, and end-point of the study.