Immune diseases caused by mutations in kinases and components of the ubiquitin system
- PMID: 24840983
- PMCID: PMC5793992
- DOI: 10.1038/ni.2892
Immune diseases caused by mutations in kinases and components of the ubiquitin system
Abstract
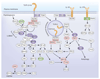
The signaling networks that control the immune system are coordinated by a myriad of interconnecting phosphorylation and ubiquitylation events. This review provides an overview of mutations in human genes encoding these proteins that give rise to immune diseases. Analysis of the biological effects of these mutations has revealed the true physiological roles of particular signaling networks and promises to revolutionize the treatment of these diseases.
Conflict of interest statement
The author declares no competing financial interests.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

