Screening of an FDA-approved compound library identifies four small-molecule inhibitors of Middle East respiratory syndrome coronavirus replication in cell culture
- PMID: 24841269
- PMCID: PMC4136071
- DOI: 10.1128/AAC.03011-14
Screening of an FDA-approved compound library identifies four small-molecule inhibitors of Middle East respiratory syndrome coronavirus replication in cell culture
Abstract
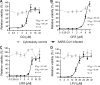
Coronaviruses can cause respiratory and enteric disease in a wide variety of human and animal hosts. The 2003 outbreak of severe acute respiratory syndrome (SARS) first demonstrated the potentially lethal consequences of zoonotic coronavirus infections in humans. In 2012, a similar previously unknown coronavirus emerged, Middle East respiratory syndrome coronavirus (MERS-CoV), thus far causing over 650 laboratory-confirmed infections, with an unexplained steep rise in the number of cases being recorded over recent months. The human MERS fatality rate of ∼ 30% is alarmingly high, even though many deaths were associated with underlying medical conditions. Registered therapeutics for the treatment of coronavirus infections are not available. Moreover, the pace of drug development and registration for human use is generally incompatible with strategies to combat emerging infectious diseases. Therefore, we have screened a library of 348 FDA-approved drugs for anti-MERS-CoV activity in cell culture. If such compounds proved sufficiently potent, their efficacy might be directly assessed in MERS patients. We identified four compounds (chloroquine, chlorpromazine, loperamide, and lopinavir) inhibiting MERS-CoV replication in the low-micromolar range (50% effective concentrations [EC(50)s], 3 to 8 μM). Moreover, these compounds also inhibit the replication of SARS coronavirus and human coronavirus 229E. Although their protective activity (alone or in combination) remains to be assessed in animal models, our findings may offer a starting point for treatment of patients infected with zoonotic coronaviruses like MERS-CoV. Although they may not necessarily reduce viral replication to very low levels, a moderate viral load reduction may create a window during which to mount a protective immune response.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures



References
-
- van Boheemen S, de Graaf M, Lauber C, Bestebroer TM, Raj VS, Zaki AM, Osterhaus AD, Haagmans BL, Gorbalenya AE, Snijder EJ, Fouchier RA. 2012. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. mBio 3:e00473-12. 10.1128/mBio.00473-12 - DOI - PMC - PubMed
-
- de Groot RJ, Baker SC, Baric RS, Brown CS, Drosten C, Enjuanes L, Fouchier RA, Galiano M, Gorbalenya AE, Memish ZA, Perlman S, Poon LL, Snijder EJ, Stephens GM, Woo PC, Zaki AM, Zambon M, Ziebuhr J. 2013. Middle East respiratory syndrome coronavirus (MERS-CoV): announcement of the Coronavirus Study Group. J. Virol. 87:7790–7792. 10.1128/JVI.01244-13 - DOI - PMC - PubMed
-
- Cotten M, Watson SJ, Zumla AI, Makhdoom HQ, Palser AL, Ong SH, Al Rabeeah AA, Alhakeem RF, Assiri A, Al-Tawfiq JA, Albarrak A, Barry M, Shibl A, Alrabiah FA, Hajjar S, Balkhy HH, Flemban H, Rambaut A, Kellam P, Memish ZA. 2014. Spread, circulation, and evolution of the Middle East respiratory syndrome coronavirus. mBio 5(1):e01062-13. 10.1128/mBio.01062-13 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

