Mechanical loading in osteocytes induces formation of a Src/Pyk2/MBD2 complex that suppresses anabolic gene expression
- PMID: 24841674
- PMCID: PMC4026426
- DOI: 10.1371/journal.pone.0097942
Mechanical loading in osteocytes induces formation of a Src/Pyk2/MBD2 complex that suppresses anabolic gene expression
Abstract
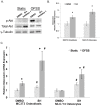
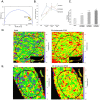
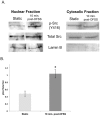
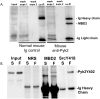
Mechanical stimulation of the skeleton promotes bone gain and suppresses bone loss, ultimately resulting in improved bone strength and fracture resistance. The molecular mechanisms directing anabolic and/or anti-catabolic actions on the skeleton during loading are not fully understood. Identifying molecular mechanisms of mechanotransduction (MTD) signaling cascades could identify new therapeutic targets. Most research into MTD mechanisms is typically focused on understanding the signaling pathways that stimulate new bone formation in response to load. However, we investigated the structural, signaling and transcriptional molecules that suppress the stimulatory effects of loading. The high bone mass phenotype of mice with global deletion of either Pyk2 or Src suggests a role for these tyrosine kinases in repression of bone formation. We used fluid shear stress as a MTD stimulus to identify a novel Pyk2/Src-mediated MTD pathway that represses mechanically-induced bone formation. Our results suggest Pyk2 and Src function as molecular switches that inhibit MTD in our mechanically stimulated osteocyte culture experiments. Once activated by oscillatory fluid shear stress (OFSS), Pyk2 and Src translocate to and accumulate in the nucleus, where they associate with a protein involved in DNA methylation and the interpretation of DNA methylation patterns -methyl-CpG-binding domain protein 2 (MBD2). OFSS-induced Cox-2 and osteopontin expression was enhanced in Pyk2 KO osteoblasts, while inhibition of Src enhanced osteocalcin expression in response to OFSS. We found that Src kinase activity increased in the nucleus of osteocytes in response to OFSS and an interaction activated between Src (Y418) and Pyk2 (Y402) increased in response to OFSS. Thus, as a mechanism to prevent an over-reaction to physical stimulation, mechanical loading may induce the formation of a Src/Pyk2/MBD2 complex in the nucleus that functions to suppress anabolic gene expression.
Conflict of interest statement
Figures


Similar articles
-
Direct visualization by FRET-FLIM of a putative mechanosome complex involving Src, Pyk2 and MBD2 in living MLO-Y4 cells.PLoS One. 2021 Dec 23;16(12):e0261660. doi: 10.1371/journal.pone.0261660. eCollection 2021. PLoS One. 2021. PMID: 34941939 Free PMC article.
-
Non-overlapping functions for Pyk2 and FAK in osteoblasts during fluid shear stress-induced mechanotransduction.PLoS One. 2011 Jan 25;6(1):e16026. doi: 10.1371/journal.pone.0016026. PLoS One. 2011. PMID: 21283581 Free PMC article.
-
Bone marrow mechanotransduction in porcine explants alters kinase activation and enhances trabecular bone formation in the absence of osteocyte signaling.Bone. 2018 Feb;107:78-87. doi: 10.1016/j.bone.2017.11.007. Epub 2017 Nov 14. Bone. 2018. PMID: 29154967
-
Nitric oxide signaling in mechanical adaptation of bone.Osteoporos Int. 2014 May;25(5):1427-37. doi: 10.1007/s00198-013-2590-4. Epub 2013 Dec 10. Osteoporos Int. 2014. PMID: 24322479 Review.
-
Cx43 and mechanotransduction in bone.Curr Osteoporos Rep. 2015 Apr;13(2):67-72. doi: 10.1007/s11914-015-0255-2. Curr Osteoporos Rep. 2015. PMID: 25616771 Free PMC article. Review.
Cited by
-
Epigenetics of Skeletal Diseases.Curr Osteoporos Rep. 2018 Jun;16(3):246-255. doi: 10.1007/s11914-018-0435-y. Curr Osteoporos Rep. 2018. PMID: 29574535 Review.
-
Pyk2 Regulates MAMs and Mitochondrial Dynamics in Hippocampal Neurons.Cells. 2022 Mar 1;11(5):842. doi: 10.3390/cells11050842. Cells. 2022. PMID: 35269464 Free PMC article.
-
The Influence of DNA Methylation on Bone Cells.Curr Genomics. 2015 Dec;16(6):384-92. doi: 10.2174/1389202916666150817202913. Curr Genomics. 2015. PMID: 27019613 Free PMC article.
-
Functionalization of Electrospun Polycaprolactone Scaffolds with Matrix-Binding Osteocyte-Derived Extracellular Vesicles Promotes Osteoblastic Differentiation and Mineralization.Ann Biomed Eng. 2021 Dec;49(12):3621-3635. doi: 10.1007/s10439-021-02872-2. Epub 2021 Oct 18. Ann Biomed Eng. 2021. PMID: 34664147 Free PMC article.
-
PIE-FLIM Measurements of Two Different FRET-Based Biosensor Activities in the Same Living Cells.Biophys J. 2020 Apr 21;118(8):1820-1829. doi: 10.1016/j.bpj.2020.03.003. Epub 2020 Mar 10. Biophys J. 2020. PMID: 32191861 Free PMC article.
References
-
- Boutahar N, Guignandon A, Vico L, Lafage-Proust MH (2004) Mechanical strain on osteoblasts activates autophosphorylation of focal adhesion kinase and proline-rich tyrosine kinase 2 tyrosine sites involved in ERK activation. The Journal of biological chemistry 279: 30588–30599. - PubMed
-
- Guignandon A, Boutahar N, Rattner A, Vico L, Lafage-Proust MH (2006) Cyclic strain promotes shuttling of PYK2/Hic-5 complex from focal contacts in osteoblast-like cells. Biochemical and biophysical research communications 343: 407–414. - PubMed
-
- Avraham H, Park SY, Schinkmann K, Avraham S (2000) RAFTK/Pyk2-mediated cellular signalling. Cellular signalling 12: 123–133. - PubMed
-
- Hall JE, Fu W, Schaller MD (2011) Focal adhesion kinase: exploring Fak structure to gain insight into function. International review of cell and molecular biology 288: 185–225. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

