Ultrasound-guided tissue fractionation by high intensity focused ultrasound in an in vivo porcine liver model
- PMID: 24843132
- PMCID: PMC4050569
- DOI: 10.1073/pnas.1318355111
Ultrasound-guided tissue fractionation by high intensity focused ultrasound in an in vivo porcine liver model
Abstract
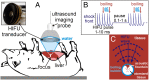
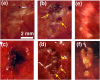
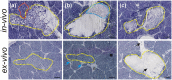
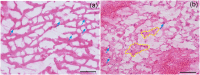
The clinical use of high intensity focused ultrasound (HIFU) therapy for noninvasive tissue ablation has been recently gaining momentum. In HIFU, ultrasound energy from an extracorporeal source is focused within the body to ablate tissue at the focus while leaving the surrounding organs and tissues unaffected. Most HIFU therapies are designed to use heating effects resulting from the absorption of ultrasound by tissue to create a thermally coagulated treatment volume. Although this approach is often successful, it has its limitations, such as the heat sink effect caused by the presence of a large blood vessel near the treatment area or heating of the ribs in the transcostal applications. HIFU-induced bubbles provide an alternative means to destroy the target tissue by mechanical disruption or, at its extreme, local fractionation of tissue within the focal region. Here, we demonstrate the feasibility of a recently developed approach to HIFU-induced ultrasound-guided tissue fractionation in an in vivo pig model. In this approach, termed boiling histotripsy, a millimeter-sized boiling bubble is generated by ultrasound and further interacts with the ultrasound field to fractionate porcine liver tissue into subcellular debris without inducing further thermal effects. Tissue selectivity, demonstrated by boiling histotripsy, allows for the treatment of tissue immediately adjacent to major blood vessels and other connective tissue structures. Furthermore, boiling histotripsy would benefit the clinical applications, in which it is important to accelerate resorption or passage of the ablated tissue volume, diminish pressure on the surrounding organs that causes discomfort, or insert openings between tissues.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Control of the dynamics of a boiling vapour bubble using pressure-modulated high intensity focused ultrasound without the shock scattering effect: A first proof-of-concept study.Ultrason Sonochem. 2021 Sep;77:105699. doi: 10.1016/j.ultsonch.2021.105699. Epub 2021 Jul 31. Ultrason Sonochem. 2021. PMID: 34371476 Free PMC article.
-
Ultrasonic atomization of tissue and its role in tissue fractionation by high intensity focused ultrasound.Phys Med Biol. 2012 Dec 7;57(23):8061-78. doi: 10.1088/0031-9155/57/23/8061. Epub 2012 Nov 16. Phys Med Biol. 2012. PMID: 23159812 Free PMC article.
-
Bubble dynamics in boiling histotripsy.Ultrasound Med Biol. 2018 Dec;44(12):2673-2696. doi: 10.1016/j.ultrasmedbio.2018.07.025. Epub 2018 Sep 15. Ultrasound Med Biol. 2018. PMID: 30228043
-
Histotripsy methods in mechanical disintegration of tissue: towards clinical applications.Int J Hyperthermia. 2015 Mar;31(2):145-62. doi: 10.3109/02656736.2015.1007538. Epub 2015 Feb 24. Int J Hyperthermia. 2015. PMID: 25707817 Free PMC article. Review.
-
Histotripsy: A Method for Mechanical Tissue Ablation with Ultrasound.Annu Rev Biomed Eng. 2024 Jul;26(1):141-167. doi: 10.1146/annurev-bioeng-073123-022334. Epub 2024 Jun 20. Annu Rev Biomed Eng. 2024. PMID: 38346277 Free PMC article. Review.
Cited by
-
Non-Invasive Ultrasound Liver Ablation Using Histotripsy: Chronic Study in an In Vivo Rodent Model.Ultrasound Med Biol. 2016 Aug;42(8):1890-902. doi: 10.1016/j.ultrasmedbio.2016.03.018. Epub 2016 Apr 29. Ultrasound Med Biol. 2016. PMID: 27140521 Free PMC article.
-
Boiling histotripsy lesion characterization on a clinical magnetic resonance imaging-guided high intensity focused ultrasound system.PLoS One. 2017 Mar 16;12(3):e0173867. doi: 10.1371/journal.pone.0173867. eCollection 2017. PLoS One. 2017. PMID: 28301597 Free PMC article.
-
Magnetic Resonance Imaging monitoring of histotripsy effects in agar phantom.Med Phys. 2025 Aug;52(8):e18054. doi: 10.1002/mp.18054. Med Phys. 2025. PMID: 40804776 Free PMC article.
-
Release of Cell-free MicroRNA Tumor Biomarkers into the Blood Circulation with Pulsed Focused Ultrasound: A Noninvasive, Anatomically Localized, Molecular Liquid Biopsy.Radiology. 2017 Apr;283(1):158-167. doi: 10.1148/radiol.2016160024. Epub 2016 Nov 1. Radiology. 2017. PMID: 27802108 Free PMC article.
-
Clot Degradation Under the Action of Histotripsy Bubble Activity and a Lytic Drug.IEEE Trans Ultrason Ferroelectr Freq Control. 2021 Sep;68(9):2942-2952. doi: 10.1109/TUFFC.2021.3052393. Epub 2021 Aug 27. IEEE Trans Ultrason Ferroelectr Freq Control. 2021. PMID: 33460375 Free PMC article.
References
-
- Dubinsky TJ, Cuevas C, Dighe MK, Kolokythas O, Hwang JH. High-intensity focused ultrasound: Current potential and oncologic applications. AJR Am J Roentgenol. 2008;190(1):191–199. - PubMed
-
- Bailey MR, Khokhlova VA, Sapozhnikov OA, Kargl SG, Crum LA. Physical mechanisms of the therapeutic effect of ultrasound (a review) Acoust Phys. 2003;49:369–388.
-
- Crouzet S, et al. Whole-gland ablation of localized prostate cancer with high-intensity focused ultrasound: Oncologic outcomes and morbidity in 1002 patients. Eur Urol. 2014;65(5):907–914. - PubMed
-
- Ren XL, et al. Extracorporeal ablation of uterine fibroids with high-intensity focused ultrasound: Imaging and histopathologic evaluation. J Ultrasound Med. 2007;26(2):201–212. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

