Moving Fe2+ from ferritin ion channels to catalytic OH centers depends on conserved protein cage carboxylates
- PMID: 24843174
- PMCID: PMC4050572
- DOI: 10.1073/pnas.1318417111
Moving Fe2+ from ferritin ion channels to catalytic OH centers depends on conserved protein cage carboxylates
Abstract
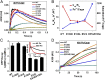
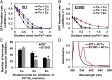
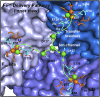
Ferritin biominerals are protein-caged metabolic iron concentrates used for iron-protein cofactors and oxidant protection (Fe(2+) and O2 sequestration). Fe(2+) passage through ion channels in the protein cages, like membrane ion channels, required for ferritin biomineral synthesis, is followed by Fe(2+) substrate movement to ferritin enzyme (Fox) sites. Fe(2+) and O2 substrates are coupled via a diferric peroxo (DFP) intermediate, λmax 650 nm, which decays to [Fe(3+)-O-Fe(3+)] precursors of caged ferritin biominerals. Structural studies show multiple conformations for conserved, carboxylate residues E136 and E57, which are between ferritin ion channel exits and enzymatic sites, suggesting functional connections. Here we show that E136 and E57 are required for ferritin enzyme activity and thus are functional links between ferritin ion channels and enzymatic sites. DFP formation (Kcat and kcat/Km), DFP decay, and protein-caged hydrated ferric oxide accumulation decreased in ferritin E57A and E136A; saturation required higher Fe(2+) concentrations. Divalent cations (both ion channel and intracage binding) selectively inhibit ferritin enzyme activity (block Fe(2+) access), Mn(2+) << Co(2+) < Cu(2+) < Zn(2+), reflecting metal ion-protein binding stabilities. Fe(2+)-Cys126 binding in ferritin ion channels, observed as Cu(2+)-S-Cys126 charge-transfer bands in ferritin E130D UV-vis spectra and resistance to Cu(2+) inhibition in ferritin C126S, was unpredicted. Identifying E57 and E136 links in Fe(2+) movement from ferritin ion channels to ferritin enzyme sites completes a bucket brigade that moves external Fe(2+) into ferritin enzymatic sites. The results clarify Fe(2+) transport within ferritin and model molecular links between membrane ion channels and cytoplasmic destinations.
Keywords: BioIron; antioxidant; ferrihydrite; iron traffic; oxidoreductase enzyme activity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Solving Biology's Iron Chemistry Problem with Ferritin Protein Nanocages.Acc Chem Res. 2016 May 17;49(5):784-91. doi: 10.1021/ar500469e. Epub 2016 May 2. Acc Chem Res. 2016. PMID: 27136423
-
Ferritin ion channel disorder inhibits Fe(II)/O2 reactivity at distant sites.Inorg Chem. 2012 Nov 5;51(21):11406-11. doi: 10.1021/ic3010135. Epub 2012 Oct 23. Inorg Chem. 2012. PMID: 23092300 Free PMC article.
-
Fe(2+) substrate transport through ferritin protein cage ion channels influences enzyme activity and biomineralization.J Biol Inorg Chem. 2015 Sep;20(6):957-69. doi: 10.1007/s00775-015-1279-x. Epub 2015 Jul 23. J Biol Inorg Chem. 2015. PMID: 26202907 Free PMC article.
-
Ferritin protein nanocages use ion channels, catalytic sites, and nucleation channels to manage iron/oxygen chemistry.Curr Opin Chem Biol. 2011 Apr;15(2):304-11. doi: 10.1016/j.cbpa.2011.01.004. Epub 2011 Feb 4. Curr Opin Chem Biol. 2011. PMID: 21296609 Free PMC article. Review.
-
Ferritin: the protein nanocage and iron biomineral in health and in disease.Inorg Chem. 2013 Nov 4;52(21):12223-33. doi: 10.1021/ic400484n. Epub 2013 Oct 8. Inorg Chem. 2013. PMID: 24102308 Free PMC article. Review.
Cited by
-
Chemistry at the protein-mineral interface in L-ferritin assists the assembly of a functional (μ3-oxo)Tris[(μ2-peroxo)] triiron(III) cluster.Proc Natl Acad Sci U S A. 2017 Mar 7;114(10):2580-2585. doi: 10.1073/pnas.1614302114. Epub 2017 Feb 15. Proc Natl Acad Sci U S A. 2017. PMID: 28202724 Free PMC article.
-
Ferritins: furnishing proteins with iron.J Biol Inorg Chem. 2016 Mar;21(1):13-28. doi: 10.1007/s00775-016-1336-0. Epub 2016 Jan 29. J Biol Inorg Chem. 2016. PMID: 26825805 Free PMC article. Review.
-
Neuroferritinopathy: From ferritin structure modification to pathogenetic mechanism.Neurobiol Dis. 2015 Sep;81:134-43. doi: 10.1016/j.nbd.2015.02.007. Epub 2015 Mar 12. Neurobiol Dis. 2015. PMID: 25772441 Free PMC article. Review.
-
Structural Insights Into the Effects of Interactions With Iron and Copper Ions on Ferritin From the Blood Clam Tegillarca granosa.Front Mol Biosci. 2022 Mar 11;9:800008. doi: 10.3389/fmolb.2022.800008. eCollection 2022. Front Mol Biosci. 2022. PMID: 35359603 Free PMC article.
-
Iron redox pathway revealed in ferritin via electron transfer analysis.Sci Rep. 2020 Mar 4;10(1):4033. doi: 10.1038/s41598-020-60640-z. Sci Rep. 2020. PMID: 32132578 Free PMC article.
References
-
- Gouaux E, Mackinnon R. Principles of selective ion transport in channels and pumps. Science. 2005;310(5753):1461–1465. - PubMed
-
- Richards LA, Schäfer AI, Richards BS, Corry B. The importance of dehydration in determining ion transport in narrow pores. Small. 2012;8(11):1701–1709. - PubMed
-
- Corringer PJ, et al. Structure and pharmacology of pentameric receptor channels: From bacteria to brain. Structure. 2012;20(6):941–956. - PubMed
-
- Liu X, Theil EC. Ferritins: Dynamic management of biological iron and oxygen chemistry. Acc Chem Res. 2005;38(3):167–175. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

