Mechanism of diabetic neuropathy: Where are we now and where to go?
- PMID: 24843457
- PMCID: PMC4008011
- DOI: 10.1111/j.2040-1124.2010.00070.x
Mechanism of diabetic neuropathy: Where are we now and where to go?
Abstract
Neuropathy is the most common complication of diabetes. As a consequence of longstanding hyperglycemia, a downstream metabolic cascade leads to peripheral nerve injury through an increased flux of the polyol pathway, enhanced advanced glycation end-products formation, excessive release of cytokines, activation of protein kinase C and exaggerated oxidative stress, as well as other confounding factors. Although these metabolic aberrations are deemed as the main stream for the pathogenesis of diabetic microvascular complications, organ-specific histological and biochemical characteristics constitute distinct mechanistic processes of neuropathy different from retinopathy or nephropathy. Extremely long axons originating in the small neuronal body are vulnerable on the most distal side as a result of malnutritional axonal support or environmental insults. Sparse vascular supply with impaired autoregulation is likely to cause hypoxic damage in the nerve. Such dual influences exerted by long-term hyperglycemia are critical for peripheral nerve damage, resulting in distal-predominant nerve fiber degeneration. More recently, cellular factors derived from the bone marrow also appear to have a strong impact on the development of peripheral nerve pathology. As evident from such complicated processes, inhibition of single metabolic factors might not be sufficient for the treatment of neuropathy, but a combination of several inhibitors might be a promising approach to overcome this serious disorder. (J Diabetes Invest, doi: 10.1111/j.2040-1124.2010.00070.x, 2010).
Keywords: Diabetic neuropathy; Novel treatment; Pathogenesis.
Figures
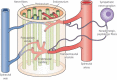



 ). However, because mitochondria are impaired to produce ATP as a result of oxygen depletion, surplus glucose enters the collateral pathway to sorbitol and phosphatidic acid. From the former, aldose reductase is activated to cause glutathione deficiency and redox deviation, as in the hyperglycemic condition (
). However, because mitochondria are impaired to produce ATP as a result of oxygen depletion, surplus glucose enters the collateral pathway to sorbitol and phosphatidic acid. From the former, aldose reductase is activated to cause glutathione deficiency and redox deviation, as in the hyperglycemic condition ( ). As a result, free radical injury and protein kinase C (PKC) activation ensue to aggravate ischemic injury (
). As a result, free radical injury and protein kinase C (PKC) activation ensue to aggravate ischemic injury ( ). Once reperfusion starts, oxygen radicals accumulate aldehydes, which are also substrates of aldose reductase, and enhance radical injury (
). Once reperfusion starts, oxygen radicals accumulate aldehydes, which are also substrates of aldose reductase, and enhance radical injury ( ) (adapted from reference 69 and modified by the author).
) (adapted from reference 69 and modified by the author).


References
-
- Boulton AJ, Vinik AI, Arezzo JC, et al. Diabetic neuropathies: a statement by the American Diabetes Association. Diabetes Care 2005; 28: 956–962 - PubMed
-
- Pirart J. Diabetes mellitus and its degenerative complications: a prospective study of 4400 patients observed. Diabetes Care 1978; 1: 168–188
-
- Vinik AI, Liuzze FJ, Holland MT, et al. Diabetic neuropathies. Diabetes Care 1992; 15: 1926–1975 - PubMed
-
- Ewing DJ, Campbell IW, Clarke BF. Mortality in diabetic autonomic neuropathy. Lancet 1976; 1: 601–603 - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

