Zolmitriptan for acute migraine attacks in adults
- PMID: 24848613
- PMCID: PMC6485805
- DOI: 10.1002/14651858.CD008616.pub2
Zolmitriptan for acute migraine attacks in adults
Abstract
Background: Migraine is a common, disabling condition and a burden for the individual, health services, and society. Zolmitriptan is an abortive medication for migraine attacks, belonging to the triptan family. These medicines work in a different way to analgesics such as paracetamol and ibuprofen.
Objectives: To determine the efficacy and tolerability of zolmitriptan compared to placebo and other active interventions in the treatment of acute migraine attacks in adults.
Search methods: We searched the Cochrane Central Register of Controlled Trials (CENTRAL) in The Cochrane Library, MEDLINE, EMBASE, and the Oxford Pain Relief Database, together with three online databases (www.astrazenecaclinicaltrials.com, www.clinicaltrials.gov, and apps.who.int/trialsearch) for studies to 12 March 2014. We also searched the reference lists of included studies and relevant reviews.
Selection criteria: We included randomised, double-blind, placebo- or active-controlled studies, with at least 10 participants per treatment arm, using zolmitriptan to treat a migraine headache episode.
Data collection and analysis: Two review authors independently assessed trial quality and extracted data. We used numbers of participants achieving each outcome to calculate risk ratios and numbers needed to treat for an additional beneficial effect (NNT) or harmful effect (NNH) compared with placebo or a different active treatment.
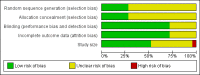
Main results: Twenty-five studies (20,162 participants) compared zolmitriptan with placebo or an active comparator. The evidence from placebo-controlled studies was of high quality for all outcomes except 24 hour outcomes and serious adverse events where only limited data were available. The majority of included studies were at a low risk of performance, detection and attrition biases, but did not adequately describe methods of randomisation and concealment.Most of the data were for the 2.5 mg and 5 mg doses compared with placebo, for treatment of moderate to severe pain. For all efficacy outcomes, zolmitriptan surpassed placebo. For oral zolmitriptan 2.5 mg versus placebo, the NNTs were 5.0, 3.2, 7.7, and 4.1 for pain-free at two hours, headache relief at two hours, sustained pain-free during the 24 hours postdose, and sustained headache relief during the 24 hours postdose, respectively. Results for the oral 5 mg dose were similar to the 2.5 mg dose, while zolmitriptan 10 mg was significantly more effective than 5 mg for pain-free and headache relief at two hours. For headache relief at one and two hours and sustained headache relief during the 24 hours postdose, but not pain-free at two hours, zolmitriptan 5 mg nasal spray was significantly more effective than the 5 mg oral tablet.For the most part, adverse events were transient and mild and were more common with zolmitriptan than placebo, with a clear dose response relationship (1 mg to 10 mg).High quality evidence from two studies showed that oral zolmitriptan 2.5 mg and 5 mg provided headache relief at two hours to the same proportion of people as oral sumatriptan 50 mg (66%, 67%, and 68% respectively), although not necessarily the same individuals. There was no significant difference in numbers experiencing adverse events. Single studies reported on other active treatment comparisons but are not described further because of the small amount of data.
Authors' conclusions: Zolmitriptan is effective as an abortive treatment for migraine attacks for some people, but is associated with increased adverse events compared to placebo. Zolmitriptan 2.5 mg and 5 mg benefited the same proportion of people as sumatriptan 50 mg, although not necessarily the same individuals, for headache relief at two hours.
Conflict of interest statement
RAM and SD have received research support from charities, government and industry sources at various times, none of which were related to this review. RAM has consulted for various pharmaceutical companies (including AstraZeneca, the manufacturers of zolmitriptan) and has received lecture fees from other pharmaceutical companies related to analgesics and other healthcare interventions, none of which were related to this review. SB has no interests to declare. AstraZeneca were not in any way involved in carrying out this review.
Figures





Update of
- doi: 10.1002/14651858.CD008616
Similar articles
-
Sumatriptan (oral route of administration) for acute migraine attacks in adults.Cochrane Database Syst Rev. 2012 Feb 15;2012(2):CD008615. doi: 10.1002/14651858.CD008615.pub2. Cochrane Database Syst Rev. 2012. PMID: 22336849 Free PMC article.
-
Sumatriptan plus naproxen for the treatment of acute migraine attacks in adults.Cochrane Database Syst Rev. 2016 Apr 20;4(4):CD008541. doi: 10.1002/14651858.CD008541.pub3. Cochrane Database Syst Rev. 2016. PMID: 27096438 Free PMC article.
-
Sumatriptan (rectal route of administration) for acute migraine attacks in adults.Cochrane Database Syst Rev. 2012 Feb 15;2012(2):CD009664. doi: 10.1002/14651858.CD009664. Cochrane Database Syst Rev. 2012. PMID: 22336868 Free PMC article.
-
Sumatriptan (subcutaneous route of administration) for acute migraine attacks in adults.Cochrane Database Syst Rev. 2012 Feb 15;2012(2):CD009665. doi: 10.1002/14651858.CD009665. Cochrane Database Syst Rev. 2012. PMID: 22336869 Free PMC article.
-
Sumatriptan plus naproxen for acute migraine attacks in adults.Cochrane Database Syst Rev. 2013 Oct 21;(10):CD008541. doi: 10.1002/14651858.CD008541.pub2. Cochrane Database Syst Rev. 2013. Update in: Cochrane Database Syst Rev. 2016 Apr 20;4:CD008541. doi: 10.1002/14651858.CD008541.pub3. PMID: 24142431 Updated.
Cited by
-
Recent advances in migraine therapy.Springerplus. 2016 May 17;5:637. doi: 10.1186/s40064-016-2211-8. eCollection 2016. Springerplus. 2016. PMID: 27330903 Free PMC article. Review.
-
Drug therapy in headache.Clin Med (Lond). 2015 Jun;15(3):273-9. doi: 10.7861/clinmedicine.15-3-273. Clin Med (Lond). 2015. PMID: 26031979 Free PMC article. Review.
-
Novel capsaicin-induced parameters of microcirculation in migraine patients revealed by imaging photoplethysmography.J Headache Pain. 2018 Jun 18;19(1):43. doi: 10.1186/s10194-018-0872-0. J Headache Pain. 2018. PMID: 29915934 Free PMC article.
-
Managing migraine with over-the-counter provision of triptans: the perspectives and readiness of Western Australian community pharmacists.PeerJ. 2019 Dec 16;7:e8134. doi: 10.7717/peerj.8134. eCollection 2019. PeerJ. 2019. PMID: 31871835 Free PMC article.
-
Why is the therapeutic effect of acute antimigraine drugs delayed? A review of controlled trials and hypotheses about the delay of effect.Br J Clin Pharmacol. 2019 Nov;85(11):2487-2498. doi: 10.1111/bcp.14090. Epub 2019 Sep 4. Br J Clin Pharmacol. 2019. PMID: 31389059 Free PMC article. Review.
References
References to studies included in this review
311CIL/0099 2000 {published data only}
-
- Astra Zeneca. A multicentre, randomised, double‐blind trial to compare the efficacy and safety of ZOMIG 2.5 mg, NARAMIG 2.5 mg and placebo in the acute treatment of adult patients with migraine. www.astrazenecaclinicaltrials.com 2005.
Charlesworth 2003 {published data only}
-
- Charlesworth BR, Dowson AJ, Purdy A, Becker WJ, Boes‐Hansen S, Färkkilä M. Speed of onset and efficacy of zolmitriptan nasal spray in the acute treatment of migraine: a randomised, double‐blind, placebo‐controlled, dose‐ranging study versus zolmitriptan tablet. CNS Drugs 2003;17(9):653‐67. - PubMed
Dahlof 1998 {published data only}
-
- Dahlöf C, Diener HC, Goadsby PJ, Massiou H, Olesen J, Schoenen J, et al. Zolmitriptan, a 5‐HT1B/1D receptor agonist for the acute oral treatment of migraine: a multicentre, dose‐range finding study. Eurean Journal of Neurology 1998;5(6):535‐43. - PubMed
Dib 2002 {published data only}
-
- Dib M, Massiou H, Weber M, Henry P, Garcia‐Acosta S, Bousser MG, Bi‐Profenid Migraine Study Group. Efficacy of oral ketoprofen in acute migraine: a double‐blind randomized clinical trial. Neurology 2002;58(11):1660‐5. - PubMed
Dodick 2005 {published data only}
-
- Dodick D, Brandes J, Elkind A, Mathew N, Rodichok L. Speed of onset, efficacy and tolerability of zolmitriptan nasal spray in the acute treatment of migraine: a randomised, double‐blind, placebo‐controlled study. CNS Drugs 2005;19(2):125‐36. - PubMed
Dowson 2002 {published data only}
-
- Dowson AJ, MacGregor EA, Purdy RA, Becker WJ, Green J, Levy SL. Zolmitriptan orally disintegrating tablet is effective in the acute treatment of migraine. Cephalalgia 2002;22(2):101‐6. - PubMed
Gallagher 2000 {published data only}
-
- Gallagher RM, Dennish G, Spierings EL, Chitra R. A comparative trial of zolmitriptan and sumatriptan for the acute oral treatment of migraine. Headache 2000;45(1):7‐16. - PubMed
Gawel 2005 {published data only}
-
- Gawel M, Aschoff J, May A, Charlesworth BR, REALIZE Study Team. Zolmitriptan 5 mg nasal spray: efficacy and onset of action in the acute treatment of migraine‐‐results from phase 1 of the REALIZE Study. Headache 2005;45(1):7‐16. - PubMed
Geraud 2000 {published data only}
-
- Geraud G, Olesen J, Pfaffenrath V, Tfelt‐Hansen P, Zupping R, Diener HC, et al. Comparison of the efficacy of zolmitriptan and sumatriptan: issues in migraine trial design. Cephalalgia 2000;20(1):30‐8. - PubMed
Geraud 2002 {published data only}
-
- Geraud G, Compagnon A, Rossi A, COZAM Study Group. Zolmitriptan versus a combination of acetylsalicylic acid and metoclopramide in the acute oral treatment of migraine: a double‐blind, randomised, three‐attack study. European Neurology 2002;47(2):88‐98. - PubMed
Goadsby 2007b {published data only}
-
- Goadsby PJ, Massiou H, Pascual J, Diener HC, Dahlöf CG, Mateos V. Almotriptan and zolmitriptan in the acute treatment of migraine. Acta Neurologica Scandinavica 2007;115(1):34‐40. - PubMed
Gruffyd‐Jones 2001 {published data only}
-
- Gruffyd‐Jones K, Kies B, Middleton A, Mulder LJ, Røsjø Ø, Millson DS. Zolmitriptan versus sumatriptan for the acute oral treatment of migraine: a randomized, double‐blind, international study. European Journal of Neurology 2001;8(3):237‐45. - PubMed
Ho 2008 {published data only}
-
- Dodick DW, Kost J, Assaid C, Lines C, Ho TW. Sustained pain freedom and no adverse events as an endpoint in clinical trials of acute migraine treatments: Application to patient‐level data from a trial of the CGRP receptor antagonist, telcagepant, and zolmitriptan. Cephalalgia 2011;31(3):296‐300. [DOI: 10.1177/0333102410385585] - DOI - PubMed
-
- Ho TW, Ferrari MD, Dodick DW, Galet V, Kost J, Fan X, et al. Efficacy and tolerability of MK‐0974 (telcagepant), a new oral antagonist of calcitonin gene‐related peptide receptor, compared with zolmitriptan for acute migraine: a randomised, placebo‐controlled, parallel‐treatment trial. Lancet 2008;372(9656):2115‐23. - PubMed
Klapper 2004 {published data only}
-
- Klapper J, Lucas C, Røsjø Ø, Charlesworth B, ZODIAC study group. Benefits of treating highly disabled migraine patients with zolmitriptan while pain is mild. Cephalalgia 2004;24(11):918‐24. - PubMed
Loder 2005 {published data only}
-
- Loder E, Freitag FG, Adelman J, Pearlmand S, Abu‐Shakra S. Pain‐free rates with zolmitriptan 2.5 mg ODT in the acute treatment of migraine: results of a large double‐blind placebo‐ controlled trial. Current Medical Research and Opinion 2005;21(3):381‐9. - PubMed
Pascual 2000 {published data only}
-
- Pascual J, Vega P, Diener HC, Allen C, Vrijens F, Patel K. Comparison of rizatriptan 10 mg vs. zolmitriptan 2.5 mg in the acute treatment of migraine. Rizatriptan‐Zolmitriptan Study Group. Cephalalgia 2000;20(5):455‐61. - PubMed
Rapoport 1997 {published data only}
-
- Rapoport AM, Ramadan NM, Adelman JU, Mathew NT, Elkind AH, Kudrow DB, et al. Optimizing the dose of zolmitriptan (Zomig, 311C90) for the acute treatment of migraine. A multicenter, double‐blind, placebo‐controlled, dose range‐finding study. The 017 Clinical Trial Study Group. Neurology 1997;49(5):1210‐8. - PubMed
Ryan 2000 {published data only}
-
- Ryan RE, Diamond S, Giammarco RAM, Aurora SK, Reed RC, Fletcher PE. Efficacy of zolmitriptan at early time‐points for the acute treatment of migraine and treatment recurrence. A randomised, placebo‐controlled trial. CNS Drugs 2000;13(3):215‐26.
Sakai 2002 {published data only}
-
- Sakai F, Iwata M, Tashiro K, Itoyama Y, Tsuji S, Fukuuchi Y, et al. Zolmitriptan is effective and well tolerated in Japanese patients with migraine: a dose‐response study. Cephalalgia 2002;22(5):376‐83. - PubMed
Solomon 1997 {published data only}
-
- Solomon GD, Cady RK, Klapper JA, Earl NL, Saper JR, Ramadan NM. Clinical efficacy and tolerability of 2.5 mg zolmitriptan for the acute treatment of migraine. The 042 Clinical Trial Study Group. Neurology 1997;49(5):1219‐25. - PubMed
Spierings 2004 {published data only}
-
- Spierings EL, Rapoport AM, Dodick DW, Charlesworth B. Acute treatment of migraine with zolmitriptan 5 mg orally disintegrating tablet. CNS Drugs 2004;18(15):1133‐41. - PubMed
Steiner 2003 {published data only}
-
- Steiner TJ, Diener HC, MacGregor EA, Schoenen J, Muirheads N, Sikes CR. Comparative efficacy of eletriptan and zolmitriptan in the acute treatment of migraine. Cephalalgia 2003;23(10):942‐52. - PubMed
Tuchman 2006 {published data only}
-
- Tuchman M, Hee A, Emeribe U, Silberstein S. Efficacy and tolerability of zolmitriptan oral tablet in the acute treatment of menstrual migraine. CNS Drugs 2006;20(12):1019‐26. - PubMed
Tullo 2010 {published data only}
-
- Allais G, Tullo V, Benedetto C, Zava D, Omboni S, Bussone G. Efficacy of frovatriptan in the acute treatment of menstrually related migraine: analysis of a double‐blind, randomized, multicenter, Italian, comparative study versus zolmitriptan. Neurological Sciences 2011;32 Suppl 1:S99‐104. [DOI: 10.1007/s10072-011-0547-y] - DOI - PMC - PubMed
-
- Tullo V, Allais G, Curone M, Ferrari MD, Omboni S, Benedetto C, et al. Frovatriptan versus zolmitriptan for the acute treatment of migraine with aura: a subgroup analysis of a double‐blind, randomized, multicenter, Italian study. Neurological Sciences 2012;33 Suppl 1:S61‐4. [DOI: 10.1007/s10072-012-1043-8] - DOI - PMC - PubMed
Visser 1996 {published data only}
-
- Visser WH, Klein KB, Cox RC, Jones D, Ferrari MD. 311C90, a new central and peripherally acting 5‐HT1D receptor agonist in the acute oral treatment of migraine: a double‐blind, placebo‐controlled, dose‐range finding study. Neurology 1996;46(2):522‐6. - PubMed
References to studies excluded from this review
Dowson 2003 {published data only}
-
- Dowson AJ, Charlesworth BR, Purdy A, Becker WJ, Boes‐Hansen S, Färkkilä M. Tolerability and consistency of effect of zolmitriptan nasal spray in a long‐term migraine treatment trial. CNS Drugs 2003;17(11):839‐51. - PubMed
Loder 2004 {published data only}
-
- Loder E, Silberstein SD, Abu‐Shakra S, Mueller L, Smith T. Efficacy and tolerability of oral zolmitriptan in menstrually associated migraine: a randomized, prospective, parallel‐group, double‐blind, placebo‐controlled study. Headache 2004;44(2):120‐30. - PubMed
Mauskop 1999 {published data only}
-
- Mauskop A, Farkkila M, Hering‐Hanit R, Rapoport A, Warner J. Zolmitriptan is effective for the treatment of persistent and recurrent migraine headache. Current Medical Research and Opinion 1999;15(4):282‐9. - PubMed
Tepper 1999 {published data only}
-
- Tepper SJ, Donnan GA, Dowson AJ, Bomhof MA, Elkind A, Meloche J, et al. A long‐term study to maximise migraine relief with zolmitriptan. Current Medical Research and Opinion 1999;15(4):254‐71. - PubMed
References to studies awaiting assessment
CTRI/2009/091/000196 {unpublished data only}
-
- Dr Avanti Biniwale (Principle investigator). A Clinical Trial to evaluate efficacy and safety of zolmitriptan 5 mg nasal spray in acute treatment of migraine. http://ctri.nic.in (accessed 13 November 2013) 2013. [CTRI/2009/091/000196]
Additional references
Amanzio 2009
Ayzenberg 2012
Bigal 2008
Bloudek 2012
-
- Bloudek LM, Stokes M, Buse DC, Wilcox TK, Lipton RB, Goadsby PJ, et al. Cost of healthcare for patients with migraine in five European countries: results from the International Burden of Migraine Study (IBMS). Journal of Headache and Pain 2012;13(5):361‐78. [DOI: 10.1007/s10194-012-0460-7] - DOI - PMC - PubMed
BNF 2013
-
- Zolmitriptan. British National Formulary 2013; Vol. March:Available at http://www.medicinescomplete.com/mc/bnf/current/PHP2859‐zolmitriptan.htm.
Buse 2011
Chen 2007
Collins 1997
-
- Collins SL, Moore RA, McQuay HJ. The visual analogue pain intensity scale: what is moderate pain in millimetres?. Pain 1997;72(1‐2):95‐7. - PubMed
Cook 1995
Derry 2012a
Derry 2012b
Derry 2012c
Derry 2012d
Derry 2013
Diamond 2007
-
- Diamond S, Bigal ME, Silberstein S, Loder E, Reed M, Lipton RB. Patterns of diagnosis and acute and preventive treatment for migraine in the United States: results from the American Migraine Prevalence and Prevention study. Headache 2007;47(3):355‐63. [DOI: 10.1111/j.1526-4610.2006.00631.x] - DOI - PubMed
Elbourne 2002
Ferrari 2002
Gendolla 2008
Goadsby 2007a
Hazard 2009
Higgins 2011
-
- Higgins JPT, Green S (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JPT, Altman DG, Sterne JAC editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
IHS 1988
-
- Headache Classification Committee of the International Headache Society. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia 1988;8(Suppl 7):1‐96. - PubMed
IHS 2000
-
- International Headache Society Clinical Trials Subcommittee. Guidelines for controlled trials of drugs in migraine: second edition. Cephalalgia 2000;20(9):765‐86. - PubMed
IHS 2004
-
- Headache Classification Subcommittee of the International Headache Society. The International Classification of Headache Disorders: 2nd edition. Cephalalgia 2004;24(Suppl 1):1‐160. - PubMed
IHS 2013
Jadad 1996a
-
- Jadad AR, Carroll D, Moore A, McQuay H. Developing a database of published reports of randomised clinical trials in pain research. Pain 1996;66(2‐3):239‐46. - PubMed
Jadad 1996b
-
- Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary?. Controlled Clinical Trials 1996;17(1):1‐12. - PubMed
Khan 1996
-
- Khan KS, Daya S, Jadad A. The importance of quality of primary studies in producing unbiased systematic reviews. Archives of Internal Medicine 1996;156(6):661‐6. - PubMed
Kirthi 2013
L'Abbé 1987
-
- L'Abbé KA, Detsky AS, O'Rourke K. Meta‐analysis in clinical research. Annals of Internal Medicine 1987;107(2):224‐33. - PubMed
Leonardi 2005
Linde 2012
Lipton 1999
-
- Lipton RB, Stewart WF. Acute migraine therapy: do doctors understand what patients with migraine want from therapy?. Headache 1999;39(Suppl 2):S20‐S26.
Lipton 2007
-
- Lipton RB, Bigal ME, Diamond M, Freitag F, Reed ML, AMPP Advisory Group, et al. Migraine prevalence, disease burden, and the need for preventive therapy. Neurology 2007;68(5):343‐9. - PubMed
Lucas 2006
Moore 1998
-
- Moore RA, Gavaghan D, Tramèr MR, Collins SL, McQuay HJ. Size is everything ‐ large amounts of information are needed to overcome random effects in estimating direction and magnitude of treatment effects. Pain 1998;78(3):209‐16. - PubMed
Moore 2008
-
- Moore RA, Barden J, Derry S, McQuay HJ. Managing potential publication bias. In: McQuay HJ, Kalso E, Moore RA editor(s). Systematic Reviews in Pain Research: Methodology Refined. Seattle: IASP Press, 2008:15‐24. [ISBN: 978‐0‐931092‐69‐5]
Moore 2010
-
- Moore RA, Eccleston C, Derry S, Wiffen P, Bell RF, Straube S, et al. ‘‘Evidence” in chronic pain – establishing best practice in the reporting of systematic reviews. Pain 2010;150:386–389. - PubMed
Morris 1995
Munakata 2009
Oldman 2002
PCA 2012
-
- Anonymous. Prescription cost analysis, England 2011. The NHS Information Centre, Prescribing Support Unit 2012. [ISBN: 978‐1‐84636‐685‐7]
Rabbie 2013
Radtke 2009
RevMan 2012 [Computer program]
-
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.2. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2012.
Song 2003
Steiner 2013
Stovner 2010
Thorlund 2014
Tramer 1997
Victor 2010
-
- Victor TW, Hu X, Campbell JC, Buse DC, Lipton RB. Migraine prevalence by age and sex in the United States: a life‐span study. Cephalalgia 2010;30(9):1065‐72. - PubMed
Vos 2012
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

