A systems biology approach utilizing a mouse diversity panel identifies genetic differences influencing isoniazid-induced microvesicular steatosis
- PMID: 24848797
- PMCID: PMC4168290
- DOI: 10.1093/toxsci/kfu094
A systems biology approach utilizing a mouse diversity panel identifies genetic differences influencing isoniazid-induced microvesicular steatosis
Abstract
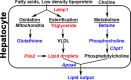
Isoniazid (INH), the mainstay therapeutic for tuberculosis infection, has been associated with rare but serious hepatotoxicity in the clinic. However, the mechanisms underlying inter-individual variability in the response to this drug have remained elusive. A genetically diverse mouse population model in combination with a systems biology approach was utilized to identify transcriptional changes, INH-responsive metabolites, and gene variants that contribute to the liver response in genetically sensitive individuals. Sensitive mouse strains developed severe microvesicular steatosis compared with corresponding vehicle control mice following 3 days of oral treatment with INH. Genes involved in mitochondrial dysfunction were enriched among liver transcripts altered with INH treatment. Those associated with INH treatment and susceptibility to INH-induced steatosis in the liver included apolipoprotein A-IV, lysosomal-associated membrane protein 1, and choline phosphotransferase 1. These alterations were accompanied by metabolomic changes including reduced levels of glutathione and the choline metabolites betaine and phosphocholine, suggesting that oxidative stress and reduced lipid export may additionally contribute to INH-induced steatosis. Finally, genome-wide association mapping revealed that polymorphisms in perilipin 2 were linked to increased triglyceride levels following INH treatment, implicating a role for inter-individual differences in lipid packaging in the susceptibility to INH-induced steatosis. Taken together, our data suggest that INH-induced steatosis is caused by not one, but multiple events involving lipid retention in the livers of genetically sensitive individuals. This work also highlights the value of using a mouse diversity panel to investigate drug-induced responses across a diverse population.
© The Author 2014. Published by Oxford University Press on behalf of the Society of Toxicology. All rights reserved. For permissions, please email: journals.permissions@oup.com.
Figures




Similar articles
-
Role of Inflammatory and Oxidative Stress, Cytochrome P450 2E1, and Bile Acid Disturbance in Rat Liver Injury Induced by Isoniazid and Lipopolysaccharide Cotreatment.Antimicrob Agents Chemother. 2016 Aug 22;60(9):5285-93. doi: 10.1128/AAC.00854-16. Print 2016 Sep. Antimicrob Agents Chemother. 2016. PMID: 27324775 Free PMC article.
-
A High Dose of Isoniazid Disturbs Endobiotic Homeostasis in Mouse Liver.Drug Metab Dispos. 2016 Nov;44(11):1742-1751. doi: 10.1124/dmd.116.070920. Epub 2016 Aug 16. Drug Metab Dispos. 2016. PMID: 27531952 Free PMC article.
-
Isoniazid-induced hepatotoxicity and neurotoxicity in rats investigated by 1H NMR based metabolomics approach.Toxicol Lett. 2018 Oct 1;295:256-269. doi: 10.1016/j.toxlet.2018.05.032. Epub 2018 Jun 21. Toxicol Lett. 2018. PMID: 29936297
-
Mechanisms of isoniazid-induced idiosyncratic liver injury: emerging role of mitochondrial stress.J Gastroenterol Hepatol. 2014 Apr;29(4):678-87. doi: 10.1111/jgh.12516. J Gastroenterol Hepatol. 2014. PMID: 24783247 Review.
-
Pharmacokinetics of isoniazid: The good, the bad, and the alternatives.Tuberculosis (Edinb). 2019 May;116S:S66-S70. doi: 10.1016/j.tube.2019.04.012. Epub 2019 Apr 26. Tuberculosis (Edinb). 2019. PMID: 31076322 Review.
Cited by
-
Systems biology for organotypic cell cultures.ALTEX. 2017;34(2):301-310. doi: 10.14573/altex.1608221. Epub 2016 Nov 14. ALTEX. 2017. PMID: 27846345 Free PMC article.
-
Identification of Candidate Risk Factor Genes for Human Idelalisib Toxicity Using a Collaborative Cross Approach.Toxicol Sci. 2019 Dec 1;172(2):265-278. doi: 10.1093/toxsci/kfz199. Toxicol Sci. 2019. PMID: 31501888 Free PMC article.
-
Genetic determinants of susceptibility to silver nanoparticle-induced acute lung inflammation in mice.FASEB J. 2017 Oct;31(10):4600-4611. doi: 10.1096/fj.201700187R. Epub 2017 Jul 17. FASEB J. 2017. PMID: 28716969 Free PMC article.
-
Disposition of intravenously or orally administered silver nanoparticles in pregnant rats and the effect on the biochemical profile in urine.J Appl Toxicol. 2017 May;37(5):530-544. doi: 10.1002/jat.3387. Epub 2016 Oct 3. J Appl Toxicol. 2017. PMID: 27696470 Free PMC article.
-
Isoniazid metabolism and hepatotoxicity.Acta Pharm Sin B. 2016 Sep;6(5):384-392. doi: 10.1016/j.apsb.2016.07.014. Epub 2016 Aug 3. Acta Pharm Sin B. 2016. PMID: 27709007 Free PMC article. Review.
References
-
- Bogue M. A., Grubb S. C. The mouse phenome project. Genetica. 2004;122:71–74. - PubMed
-
- Boelsterli U. A., Lee K. K. Mechanisms of isoniazid-induced idiosyncratic liver injury: Emerging role of mitochondrial stress. J. Gastroenterol. Hepatol. 2014;29:678–687. - PubMed
-
- Corcelle E. A., Puustinen P., Jäättelä M. Apoptosis and autophagy: Targeting autophagy signalling in cancer cells—‘trick or treats’? FEBS J. 2009;276:6084–6096. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

