IQGAP3 promotes EGFR-ERK signaling and the growth and metastasis of lung cancer cells
- PMID: 24849319
- PMCID: PMC4029748
- DOI: 10.1371/journal.pone.0097578
IQGAP3 promotes EGFR-ERK signaling and the growth and metastasis of lung cancer cells
Abstract
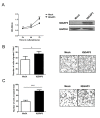
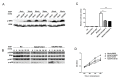
Proteins of the IQGAP family display complicated and often contradictory activities in tumorigenesis. IQGAP1 has well documented oncogenic potential and IQGAP2 has putative tumor-suppressive function. IQGAP3 is the latest addition to this family and its role in cancer development remains to be defined. Here we demonstrate IQGAP3 expression is markedly increased in lung cancer tissues at both mRNA and protein levels. Overexpression of IQGAP3 promoted tumor cell growth, and migration and invasion, whereas knockdown of IQGAP3 exhibited opposite effects. Moreover, suppression of IQGAP3 in a lung cancer cell line caused a reduction in the tumorigenicity of these cells in lung tissue after intravenous injection. Furthermore, we showed that IQGAP3 is able to interact with ERK1 and enhance its phosphorylation following treatment with EGF. These data suggest that IQGAP3 may contribute to the pathogenesis of lung cancer by modulating EGFR-ERK signaling.
Conflict of interest statement
Figures




Similar articles
-
The Antithetic Roles of IQGAP2 and IQGAP3 in Cancers.Cancers (Basel). 2023 Feb 9;15(4):1115. doi: 10.3390/cancers15041115. Cancers (Basel). 2023. PMID: 36831467 Free PMC article. Review.
-
IQGAP3 signalling mediates intratumoral functional heterogeneity to enhance malignant growth.Gut. 2025 Feb 6;74(3):364-386. doi: 10.1136/gutjnl-2023-330390. Gut. 2025. PMID: 39438124 Free PMC article.
-
Role of IQGAP3 in metastasis and epithelial-mesenchymal transition in human hepatocellular carcinoma.J Transl Med. 2017 Aug 15;15(1):176. doi: 10.1186/s12967-017-1275-8. J Transl Med. 2017. PMID: 28810875 Free PMC article.
-
Involvement of IQGAP3, a regulator of Ras/ERK-related cascade, in hepatocyte proliferation in mouse liver regeneration and development.J Cell Physiol. 2009 Sep;220(3):621-31. doi: 10.1002/jcp.21798. J Cell Physiol. 2009. PMID: 19452445
-
IQGAPs in cancer: a family of scaffold proteins underlying tumorigenesis.FEBS Lett. 2009 Jun 18;583(12):1817-24. doi: 10.1016/j.febslet.2009.05.007. Epub 2009 May 9. FEBS Lett. 2009. PMID: 19433088 Free PMC article. Review.
Cited by
-
IQ Motif Containing GTPase Activating Proteins (IQGAPs), A-Kinase Anchoring Proteins (AKAPs) and Kinase Suppressor of Ras Proteins (KSRs) in Scaffolding Oncogenic Pathways and Their Therapeutic Potential.ACS Omega. 2022 Dec 6;7(50):45837-45848. doi: 10.1021/acsomega.2c05505. eCollection 2022 Dec 20. ACS Omega. 2022. PMID: 36570181 Free PMC article. Review.
-
Molecular carcinogenesis of gastric cancer: Lauren classification, mucin phenotype expression, and cancer stem cells.Int J Clin Oncol. 2019 Jul;24(7):771-778. doi: 10.1007/s10147-019-01443-9. Epub 2019 Apr 12. Int J Clin Oncol. 2019. PMID: 30980196 Review.
-
Wnt target IQGAP3 promotes Wnt signaling via disrupting Axin1-CK1α interaction.Oncogene. 2025 Aug 19. doi: 10.1038/s41388-025-03512-y. Online ahead of print. Oncogene. 2025. PMID: 40830657
-
The Antithetic Roles of IQGAP2 and IQGAP3 in Cancers.Cancers (Basel). 2023 Feb 9;15(4):1115. doi: 10.3390/cancers15041115. Cancers (Basel). 2023. PMID: 36831467 Free PMC article. Review.
-
IQGAP3 activates Hedgehog signaling to confer stemness and metastasis via up-regulating GLI1 in lung cancer.Sci Rep. 2024 Dec 28;14(1):31327. doi: 10.1038/s41598-024-82793-x. Sci Rep. 2024. PMID: 39732803 Free PMC article.
References
-
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, et al. (2009) Cancer statistics, 2009. CA: a cancer journal for clinicians 59: 225–249. - PubMed
-
- Jemal A, Bray F (2011) Center MM, Ferlay J, Ward E, et al (2011) Global cancer statistics. CA: a cancer journal for clinicians 61: 69–90. - PubMed
-
- Houwen L (2003) State of the art:lung cancer in China. Annals of thoracic and cardiovascular surgery : official journal of the Association of Thoracic and Cardiovascular Surgeons of Asia 9: 147–148. - PubMed
-
- Koudelakova V, Kneblova M, Trojanec R, Drabek J, Hajduch M (2013) Non-small cell lung cancer - genetic predictors. Biomedical papers of the Medical Faculty of the University Palacky, Olomouc, Czechoslovakia 157: 125–136. - PubMed
-
- Ettinger DS, Akerley W, Bepler G, Blum MG, Chang A, et al. (2010) Non-small cell lung cancer. Journal of the National Comprehensive Cancer Network : JNCCN 8: 740–801. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

