Thermodynamics and mechanism of the interaction of willardiine partial agonists with a glutamate receptor: implications for drug development
- PMID: 24850223
- PMCID: PMC4215890
- DOI: 10.1021/bi500511m
Thermodynamics and mechanism of the interaction of willardiine partial agonists with a glutamate receptor: implications for drug development
Abstract
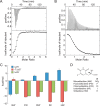
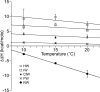
Understanding the thermodynamics of binding of a lead compound to a receptor can provide valuable information for drug design. The binding of compounds, particularly partial agonists, to subtypes of the α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid (AMPA) receptor is, in some cases, driven by increases in entropy. Using a series of partial agonists based on the structure of the natural product, willardiine, we show that the charged state of the ligand determines the enthalpic contribution to binding. Willardiines have uracil rings with pKa values ranging from 5.5 to 10. The binding of the charged form is largely driven by enthalpy, while that of the uncharged form is largely driven by entropy. This is due at least in part to changes in the hydrogen bonding network within the binding site involving one water molecule. This work illustrates the importance of charge to the thermodynamics of binding of agonists and antagonists to AMPA receptors and provides clues for further drug discovery.
Figures

Similar articles
-
A quantum biochemistry investigation of willardiine partial agonism in AMPA receptors.Phys Chem Chem Phys. 2015 May 21;17(19):13092-103. doi: 10.1039/c4cp05630b. Phys Chem Chem Phys. 2015. PMID: 25915595
-
Excitatory amino acid receptor ligands: resolution, absolute stereochemistry, and enantiopharmacology of 2-amino-3-(4-butyl-3-hydroxyisoxazol-5-yl)propionic acid.J Med Chem. 1998 Mar 12;41(6):930-9. doi: 10.1021/jm9706731. J Med Chem. 1998. PMID: 9526567
-
Probing the function, conformational plasticity, and dimer-dimer contacts of the GluR2 ligand-binding core: studies of 5-substituted willardiines and GluR2 S1S2 in the crystal.Biochemistry. 2003 May 13;42(18):5201-13. doi: 10.1021/bi020632t. Biochemistry. 2003. PMID: 12731861
-
Ligands for glutamate receptors: design and therapeutic prospects.J Med Chem. 2000 Jul 13;43(14):2609-45. doi: 10.1021/jm000007r. J Med Chem. 2000. PMID: 10893301 Review. No abstract available.
-
The AMPA receptor as a therapeutic target: current perspectives and emerging possibilities.Future Med Chem. 2010 May;2(5):877-91. doi: 10.4155/fmc.10.27. Future Med Chem. 2010. PMID: 21426207 Review.
Cited by
-
Pharmacology and Structural Analysis of Ligand Binding to the Orthosteric Site of Glutamate-Like GluD2 Receptors.Mol Pharmacol. 2016 Feb;89(2):253-62. doi: 10.1124/mol.115.100909. Epub 2015 Dec 10. Mol Pharmacol. 2016. PMID: 26661043 Free PMC article.
-
Willardiine and Its Synthetic Analogues: Biological Aspects and Implications in Peptide Chemistry of This Nucleobase Amino Acid.Pharmaceuticals (Basel). 2022 Oct 10;15(10):1243. doi: 10.3390/ph15101243. Pharmaceuticals (Basel). 2022. PMID: 36297355 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

