Lymph-borne CD8α+ dendritic cells are uniquely able to cross-prime CD8+ T cells with antigen acquired from intestinal epithelial cells
- PMID: 24850430
- PMCID: PMC4156465
- DOI: 10.1038/mi.2014.40
Lymph-borne CD8α+ dendritic cells are uniquely able to cross-prime CD8+ T cells with antigen acquired from intestinal epithelial cells
Abstract
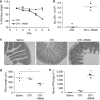
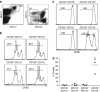
Cross-presentation of cellular antigens is crucial for priming CD8(+) T cells, and generating immunity to intracellular pathogens--particularly viruses. It is unclear which intestinal phagocytes perform this function in vivo. To address this, we examined dendritic cells (DCs) from the intestinal lymph of IFABP-tOVA 232-4 mice, which express ovalbumin in small intestinal epithelial cells (IECs). Among lymph DCs (LDCs) only CD103(+) CD11b(-) CD8α(+) DCs cross-present IEC-derived ovalbumin to CD8(+) OT-I T cells. Similarly, in the mesenteric lymph nodes (MLNs), cross-presentation of IEC-ovalbumin was limited to the CD11c(+) MHCII(hi) CD8α(+) migratory DCs, but absent from all other subsets, including the resident CD8α(hi) DCs. Crucially, delivery of purified CD8α(+) LDCs, but not other LDC subsets, into the MLN subcapsular lymphatic sinus induced proliferation of ovalbumin-specific, gut-tropic CD8(+) T cells in vivo. Finally, in 232-4 mice treated with R848, CD8α(+) LDCs were uniquely able to cross-prime interferon γ-producing CD8(+) T cells and drive their migration to the intestine. Our results clearly demonstrate that migrating CD8α(+) intestinal DCs are indispensable for cross-presentation of cellular antigens and, in conditions of inflammation, for the initial differentiation of effector CD8(+) T cells. They may therefore represent an important target for the development of antiviral vaccinations.
Figures




References
-
- Bonifaz L., Bonnyay D., Mahnke K., Rivera M., Nussenzweig M.C., Steinman R.M. Efficient targeting of protein antigen to the dendritic cell receptor DEC-205 in the steady state leads to antigen presentation on major histocompatibility complex class I products and peripheral CD8+ T cell tolerance. J. Exp. Med. 2002;196:1627–1638. - PMC - PubMed
-
- Allan R.S., et al. Migratory dendritic cells transfer antigen to a lymph node-resident dendritic cell population for efficient CTL priming. Immunity. 2006;25:153–162. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

