Cbfβ deletion in mice recapitulates cleidocranial dysplasia and reveals multiple functions of Cbfβ required for skeletal development
- PMID: 24850862
- PMCID: PMC4060659
- DOI: 10.1073/pnas.1310617111
Cbfβ deletion in mice recapitulates cleidocranial dysplasia and reveals multiple functions of Cbfβ required for skeletal development
Abstract
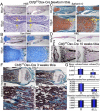
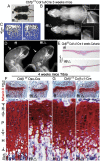
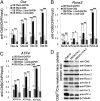
The pathogenesis of cleidocranial dysplasia (CCD) as well as the specific role of core binding factor β (Cbfβ) and the Runt-related transcription factor (RUNX)/Cbfβ complex in postnatal skeletogenesis remain unclear. We demonstrate that Cbfβ ablation in osteoblast precursors, differentiating chondrocytes, osteoblasts, and odontoblasts via Osterix-Cre, results in severe craniofacial dysplasia, skeletal dysplasia, abnormal teeth, and a phenotype recapitulating the clinical features of CCD. Cbfβ(f/f)Osterix-Cre mice have fewer proliferative and hypertrophic chondrocytes, fewer osteoblasts, and almost absent trabecular bone, indicating that Cbfβ may maintain trabecular bone formation through its function in hypertrophic chondrocytes and osteoblasts. Cbfβ(f/f)Collagen, type 1, alpha 1 (Col1α1)-Cre mice show decreased bone mineralization and skeletal deformities, but no radical deformities in teeth, mandibles, or cartilage, indicating that osteoblast lineage-specific ablation of Cbfβ results in milder bone defects and less resemblance to CCD. Activating transcription factor 4 (Atf4) and Osterix protein levels in both mutant mice are dramatically reduced. ChIP assays show that Cbfβ directly associates with the promoter regions of Atf4 and Osterix. Our data further demonstrate that Cbfβ highly up-regulates the expression of Atf4 at the transcriptional regulation level. Overall, our genetic dissection approach revealed that Cbfβ plays an indispensable role in postnatal skeletal development and homeostasis in various skeletal cell types, at least partially by up-regulating the expression of Atf4 and Osterix. It also revealed that CCD may result from functional defects of the Runx2/Cbfβ heterodimeric complex in various skeletal cells. These insights into the role of Cbfβ in postnatal skeletogenesis and CCD pathogenesis may assist in the development of new therapies for CCD and osteoporosis.
Keywords: chondrocyte differentiation; endochondral bone formation; growth plate formation; ossification; osteoblast differentiation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Otto F, et al. Cbfa1, a candidate gene for cleidocranial dysplasia syndrome, is essential for osteoblast differentiation and bone development. Cell. 1997;89(5):765–771. - PubMed
-
- Mundlos S, et al. Mutations involving the transcription factor CBFA1 cause cleidocranial dysplasia. Cell. 1997;89(5):773–779. - PubMed
-
- Lee B, et al. Missense mutations abolishing DNA binding of the osteoblast-specific transcription factor OSF2/CBFA1 in cleidocranial dysplasia. Nat Genet. 1997;16(3):307–310. - PubMed
-
- Goto T, et al. Large fontanelles are a shared feature of haploinsufficiency of RUNX2 and its co-activator CBFB. Congenit Anom (Kyoto) 2004;44(4):225–229. - PubMed
-
- Khan A, Hyde RK, Dutra A, Mohide P, Liu P. Core binding factor beta (CBFB) haploinsufficiency due to an interstitial deletion at 16q21q22 resulting in delayed cranial ossification, cleft palate, congenital heart anomalies, and feeding difficulties but favorable outcome. Am J Med Genet A. 2006;140(21):2349–2354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

