Cell types, network homeostasis, and pathological compensation from a biologically plausible ion channel expression model
- PMID: 24853940
- PMCID: PMC4109293
- DOI: 10.1016/j.neuron.2014.04.002
Cell types, network homeostasis, and pathological compensation from a biologically plausible ion channel expression model
Erratum in
- Neuron. 2015 Dec 16;88(6):1308
-
Cell Types, Network Homeostasis, and Pathological Compensation from a Biologically Plausible Ion Channel Expression Model.Neuron. 2015 Dec 16;88(6):1308. doi: 10.1016/j.neuron.2015.12.002. Epub 2015 Dec 17. Neuron. 2015. PMID: 29669236 No abstract available.
Abstract
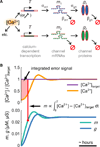
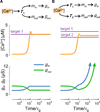
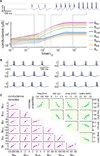
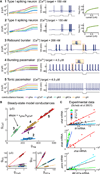
How do neurons develop, control, and maintain their electrical signaling properties in spite of ongoing protein turnover and perturbations to activity? From generic assumptions about the molecular biology underlying channel expression, we derive a simple model and show how it encodes an "activity set point" in single neurons. The model generates diverse self-regulating cell types and relates correlations in conductance expression observed in vivo to underlying channel expression rates. Synaptic as well as intrinsic conductances can be regulated to make a self-assembling central pattern generator network; thus, network-level homeostasis can emerge from cell-autonomous regulation rules. Finally, we demonstrate that the outcome of homeostatic regulation depends on the complement of ion channels expressed in cells: in some cases, loss of specific ion channels can be compensated; in others, the homeostatic mechanism itself causes pathological loss of function.
Copyright © 2014 Elsevier Inc. All rights reserved.
Figures



Comment in
-
Channeling the Central Dogma.Neuron. 2014 May 21;82(4):725-7. doi: 10.1016/j.neuron.2014.05.008. Neuron. 2014. PMID: 24853932 Free PMC article.
References
-
- Abbott L, LeMasson G. Analysis of neuron models with dynamically regulated conductances. Neural. Comput. 1993;5:823–842.
-
- Alon U. An Introduction to Systems Biology: Design Principles of Biological Circuits. Boca Raton: Chapman & Hall/CRC; 2007.
-
- Alon U, Surette MG, Barkai N, Leibler S. Robustness in bacterial chemotaxis. Nature. 1999;397:168–171. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

