Does autophagy mediate age-dependent effect of dietary restriction responses in the filamentous fungus Podospora anserina?
- PMID: 24864315
- PMCID: PMC4032521
- DOI: 10.1098/rstb.2013.0447
Does autophagy mediate age-dependent effect of dietary restriction responses in the filamentous fungus Podospora anserina?
Abstract
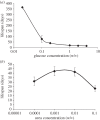
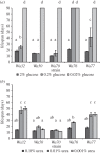
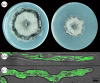
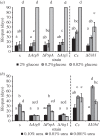
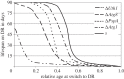
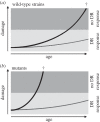
Autophagy is a well-conserved catabolic process, involving the degradation of a cell's own components through the lysosomal/vacuolar machinery. Autophagy is typically induced by nutrient starvation and has a role in nutrient recycling, cellular differentiation, degradation and programmed cell death. Another common response in eukaryotes is the extension of lifespan through dietary restriction (DR). We studied a link between DR and autophagy in the filamentous fungus Podospora anserina, a multicellular model organism for ageing studies and mitochondrial deterioration. While both carbon and nitrogen restriction extends lifespan in P. anserina, the size of the effect varied with the amount and type of restricted nutrient. Natural genetic variation for the DR response exists. Whereas a switch to carbon restriction up to halfway through the lifetime resulted in extreme lifespan extension for wild-type P. anserina, all autophagy-deficient strains had a shorter time window in which ageing could be delayed by DR. Under nitrogen limitation, only PaAtg1 and PaAtg8 mediate the effect of lifespan extension; the other autophagy-deficient mutants PaPspA and PaUth1 had a similar response as wild-type. Our results thus show that the ageing process impinges on the DR response and that this at least in part involves the genetic regulation of autophagy.
Keywords: Podospora anserina; ageing; autophagy; carbon and nitrogen restriction; dietary restriction; lifespan extension.
© 2014 The Author(s) Published by the Royal Society. All rights reserved.
Figures

Similar articles
-
The autophagy interaction network of the aging model Podospora anserina.BMC Bioinformatics. 2017 Mar 27;18(1):196. doi: 10.1186/s12859-017-1603-2. BMC Bioinformatics. 2017. PMID: 28347269 Free PMC article.
-
Identification of autophagy as a longevity-assurance mechanism in the aging model Podospora anserina.Autophagy. 2014 May;10(5):822-34. doi: 10.4161/auto.28148. Epub 2014 Feb 27. Autophagy. 2014. PMID: 24584154 Free PMC article.
-
Autophagy compensates impaired energy metabolism in CLPXP-deficient Podospora anserina strains and extends healthspan.Aging Cell. 2017 Aug;16(4):704-715. doi: 10.1111/acel.12600. Epub 2017 Apr 27. Aging Cell. 2017. PMID: 28449241 Free PMC article.
-
Calorie restriction in the filamentous fungus Podospora anserina.Exp Gerontol. 2010 Aug;45(7-8):516-24. doi: 10.1016/j.exger.2010.01.002. Epub 2010 Jan 11. Exp Gerontol. 2010. PMID: 20064602 Review.
-
Mitochondrial metabolism and aging in the filamentous fungus Podospora anserina.Biochim Biophys Acta. 2006 May-Jun;1757(5-6):604-10. doi: 10.1016/j.bbabio.2006.03.005. Epub 2006 Mar 30. Biochim Biophys Acta. 2006. PMID: 16624249 Review.
Cited by
-
Lentinula edodes Genome Survey and Postharvest Transcriptome Analysis.Appl Environ Microbiol. 2017 May 1;83(10):e02990-16. doi: 10.1128/AEM.02990-16. Print 2017 May 15. Appl Environ Microbiol. 2017. PMID: 28314725 Free PMC article.
-
Superoxide Dismutases in Eukaryotic Microorganisms: Four Case Studies.Antioxidants (Basel). 2022 Jan 19;11(2):188. doi: 10.3390/antiox11020188. Antioxidants (Basel). 2022. PMID: 35204070 Free PMC article. Review.
-
What cost mitochondria? The maintenance of functional mitochondrial DNA within and across generations.Philos Trans R Soc Lond B Biol Sci. 2014 Jul 5;369(1646):20130438. doi: 10.1098/rstb.2013.0438. Philos Trans R Soc Lond B Biol Sci. 2014. PMID: 24864309 Free PMC article. Review.
References
-
- Weindruch RH, Walford RL. 1988. The retardation of aging and disease by dietary restriction. Springfield, IL: CC Thomas.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

