Neutrophils generate microparticles during exposure to inert gases due to cytoskeletal oxidative stress
- PMID: 24867949
- PMCID: PMC4081925
- DOI: 10.1074/jbc.M113.543702
Neutrophils generate microparticles during exposure to inert gases due to cytoskeletal oxidative stress
Abstract
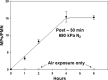
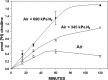
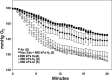
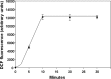
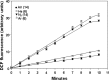
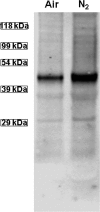
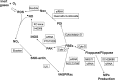
This investigation was to elucidate the mechanism for microparticle (MP) formation triggered by exposures to high pressure inert gases. Human neutrophils generate MPs at a threshold of ∼186 kilopascals with exposures of 30 min or more. Murine cells are similar, but MP production occurs at a slower rate and continues for ∼4 h, whether or not cells remain under pressure. Neutrophils exposed to elevated gas but not hydrostatic pressure produce MPs according to the potency series: argon ≃ nitrogen > helium. Following a similar pattern, gases activate type-2 nitric-oxide synthase (NOS-2) and NADPH oxidase (NOX). MP production does not occur with neutrophils exposed to a NOX inhibitor (Nox2ds) or a NOS-2 inhibitor (1400W) or with cells from mice lacking NOS-2. Reactive species cause S-nitrosylation of cytosolic actin that enhances actin polymerization. Protein cross-linking and immunoprecipitation studies indicate that increased polymerization occurs because of associations involving vasodilator-stimulated phosphoprotein, focal adhesion kinase, the H(+)/K(+) ATPase β (flippase), the hematopoietic cell multidrug resistance protein ABC transporter (floppase), and protein-disulfide isomerase in proximity to short actin filaments. Using chemical inhibitors or reducing cell concentrations of any of these proteins with small inhibitory RNA abrogates NOS-2 activation, reactive species generation, actin polymerization, and MP production. These effects were also inhibited in cells exposed to UV light, which photoreverses S-nitrosylated cysteine residues and by co-incubations with the antioxidant ebselen or cytochalasin D. The autocatalytic cycle of protein activation is initiated by inert gas-mediated singlet O2 production.
Keywords: Actin; Decompression; Focal Adhesion Kinase; NADPH Oxidase; Nitrosative Stress; Reactive Nitrogen Species (RNS); Reactive Oxygen Species (ROS); S-Nitrosylation; Singlet Oxygen.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


Similar articles
-
Neutrophil microparticle production and inflammasome activation by hyperglycemia due to cytoskeletal instability.J Biol Chem. 2017 Nov 3;292(44):18312-18324. doi: 10.1074/jbc.M117.802629. Epub 2017 Sep 25. J Biol Chem. 2017. PMID: 28972154 Free PMC article.
-
Microparticle formation by platelets exposed to high gas pressures - An oxidative stress response.Free Radic Biol Med. 2016 Dec;101:154-162. doi: 10.1016/j.freeradbiomed.2016.10.010. Epub 2016 Oct 15. Free Radic Biol Med. 2016. PMID: 27751909
-
Factors Associated with Nitric Oxide-mediated β2 Integrin Inhibition of Neutrophils.J Biol Chem. 2015 Jul 10;290(28):17474-84. doi: 10.1074/jbc.M115.651620. Epub 2015 Jun 1. J Biol Chem. 2015. PMID: 26032418 Free PMC article.
-
Ras-related GTPases and the cytoskeleton.Mol Biol Cell. 1992 May;3(5):475-9. doi: 10.1091/mbc.3.5.475. Mol Biol Cell. 1992. PMID: 1611153 Free PMC article. Review.
-
Nitric oxide synthase inhibition and oxidative stress in cardiovascular diseases: possible therapeutic targets?Pharmacol Ther. 2013 Dec;140(3):239-57. doi: 10.1016/j.pharmthera.2013.07.004. Epub 2013 Jul 13. Pharmacol Ther. 2013. PMID: 23859953 Review.
Cited by
-
Metabolic landscape in venous thrombosis: insights into molecular biology and therapeutic implications.Ann Med. 2024 Dec;56(1):2401112. doi: 10.1080/07853890.2024.2401112. Epub 2024 Sep 19. Ann Med. 2024. PMID: 39297312 Free PMC article. Review.
-
Microparticles in Human Perspiration as an Inflammatory Response Index.Diagnostics (Basel). 2024 Jun 19;14(12):1293. doi: 10.3390/diagnostics14121293. Diagnostics (Basel). 2024. PMID: 38928708 Free PMC article.
-
Plasma gelsolin modulates the production and fate of IL-1β-containing microparticles following high-pressure exposure and decompression.J Appl Physiol (1985). 2021 May 1;130(5):1604-1613. doi: 10.1152/japplphysiol.01062.2020. Epub 2021 Mar 25. J Appl Physiol (1985). 2021. PMID: 33764168 Free PMC article.
-
CNS function and dysfunction during exposure to hyperbaric oxygen in operational and clinical settings.Redox Biol. 2019 Oct;27:101159. doi: 10.1016/j.redox.2019.101159. Epub 2019 Mar 9. Redox Biol. 2019. PMID: 30902504 Free PMC article. Review.
-
Neutrophil microparticle production and inflammasome activation by hyperglycemia due to cytoskeletal instability.J Biol Chem. 2017 Nov 3;292(44):18312-18324. doi: 10.1074/jbc.M117.802629. Epub 2017 Sep 25. J Biol Chem. 2017. PMID: 28972154 Free PMC article.
References
-
- Vince R. V., McNaughton L. R., Taylor L., Midgley A. W., Laden G., Madden L. A. (2009) Release of VCAM-1 associated endothelial microparticles following simulated SCUBA dives. Eur. J. Appl. Physiol. 105, 507–513 - PubMed
-
- Madden L. A., Chrismas B. C., Mellor D., Vince R. V., Midgley A. W., McNaughton L. R., Atkin S. L., Laden G. (2010) Endothelial function and stress response after simulated dives to 18 msw breathing air or oxygen. Aviat. Space Environ. Med. 81, 41–45 - PubMed
-
- Thom S. R., Milovanova T. N., Bogush M., Bhopale V. M., Yang M., Bushmann K., Pollock N. W., Ljubkovic M., Denoble P., Dujic Z. (2012) Microparticle production, neutrophil activation and intravascular bubbles following open-water SCUBA diving. J. Appl. Physiol. 112, 1268–1278 - PubMed
-
- Thom S. R., Milovanova T. N., Bogush M., Yang M., Bhopale V. M., Pollock N. W., Ljubkovic M., Denoble P., Madden D., Lozo M., Dujic Z. (2013) Bubbles, microparticles and neutrophil activation: changes with exercise level and breathing gas during open-water SCUBA diving. J. Appl. Physiol. 114, 1396–1405 - PubMed
-
- Pontier J. M., Gempp E., Ignatescu M. (2012) Blood platelet-derived microparticles release and bubble formation after an open-sea dive. Appl. Physiol. Nutr. Metab. 37, 888–892 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

