Population pharmacokinetics and clinical response for artemether-lumefantrine in pregnant and nonpregnant women with uncomplicated Plasmodium falciparum malaria in Tanzania
- PMID: 24867986
- PMCID: PMC4136066
- DOI: 10.1128/AAC.02595-14
Population pharmacokinetics and clinical response for artemether-lumefantrine in pregnant and nonpregnant women with uncomplicated Plasmodium falciparum malaria in Tanzania
Abstract
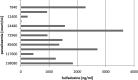
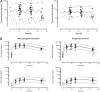
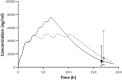
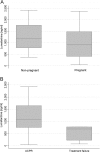
Artemether-lumefantrine (AL) is the first-line treatment for uncomplicated malaria in the second and third trimesters of pregnancy. Its efficacy during pregnancy has recently been challenged due to altered pharmacokinetic (PK) properties in this vulnerable group. The aim of this study was to determine the PK profile of AL in pregnant and nonpregnant women and assess their therapeutic outcome. Thirty-three pregnant women and 22 nonpregnant women with malaria were treated with AL (80/480 mg) twice daily for 3 days. All patients provided five venous plasma samples for drug quantification at random times over 7 days. Inter- and intraindividual variability was assessed, and the effects of covariates were quantified using a nonlinear mixed-effects modeling approach (NONMEM). A one-compartment model with first-order absorption and elimination with linear metabolism from drug to metabolite fitted the data best for both arthemether (AM) and lumefantrine (LF) and their metabolites. Pregnancy status and diarrhea showed a significant influence on LF PK. The relative bioavailability of lumefantrine and its metabolism rate into desmethyl-lumefantrine were, respectively, 34% lower and 78% higher in pregnant women than in nonpregnant patients. The overall PCR-uncorrected treatment failure rates were 18% in pregnant women and 5% in nonpregnant women (odds ratio [OR] = 4.04; P value of 0.22). A high median day 7 lumefantrine concentration was significantly associated with adequate clinical and parasitological response (P = 0.03). The observed reduction in the relative bioavailability of lumefantrine in pregnant women may explain the higher treatment failure in this group, mostly due to lower posttreatment prophylaxis. Hence, a modified treatment regimen of malaria in pregnancy should be considered.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures
Similar articles
-
Artemether-Lumefantrine Pharmacokinetics and Clinical Response Are Minimally Altered in Pregnant Ugandan Women Treated for Uncomplicated Falciparum Malaria.Antimicrob Agents Chemother. 2015 Dec 14;60(3):1274-82. doi: 10.1128/AAC.01605-15. Antimicrob Agents Chemother. 2015. PMID: 26666942 Free PMC article. Clinical Trial.
-
Population pharmacokinetics of lumefantrine in pregnant women treated with artemether-lumefantrine for uncomplicated Plasmodium falciparum malaria.Antimicrob Agents Chemother. 2009 Sep;53(9):3837-46. doi: 10.1128/AAC.00195-09. Epub 2009 Jun 29. Antimicrob Agents Chemother. 2009. PMID: 19564366 Free PMC article. Clinical Trial.
-
The pharmacokinetics of artemether and lumefantrine in pregnant women with uncomplicated falciparum malaria.Eur J Clin Pharmacol. 2006 Dec;62(12):1021-31. doi: 10.1007/s00228-006-0199-7. Epub 2006 Oct 20. Eur J Clin Pharmacol. 2006. PMID: 17053895 Clinical Trial.
-
Understanding the pharmacokinetics of Coartem.Malar J. 2009 Oct 12;8 Suppl 1(Suppl 1):S4. doi: 10.1186/1475-2875-8-S1-S4. Malar J. 2009. PMID: 19818171 Free PMC article. Review.
-
Coartemether (artemether and lumefantrine): an oral antimalarial drug.Expert Rev Anti Infect Ther. 2004 Apr;2(2):181-96. doi: 10.1586/14787210.2.2.181. Expert Rev Anti Infect Ther. 2004. PMID: 15482185 Review.
Cited by
-
Drug treatment and prevention of malaria in pregnancy: a critical review of the guidelines.Malar J. 2021 Jan 23;20(1):62. doi: 10.1186/s12936-020-03565-2. Malar J. 2021. PMID: 33485330 Free PMC article. Review.
-
Artemether-Lumefantrine Pharmacokinetics and Clinical Response Are Minimally Altered in Pregnant Ugandan Women Treated for Uncomplicated Falciparum Malaria.Antimicrob Agents Chemother. 2015 Dec 14;60(3):1274-82. doi: 10.1128/AAC.01605-15. Antimicrob Agents Chemother. 2015. PMID: 26666942 Free PMC article. Clinical Trial.
-
Effect of pharmacogenetics on plasma lumefantrine pharmacokinetics and malaria treatment outcome in pregnant women.Malar J. 2017 Jul 3;16(1):267. doi: 10.1186/s12936-017-1914-9. Malar J. 2017. PMID: 28673292 Free PMC article.
-
Population Pharmacokinetics of Artemether, Dihydroartemisinin, and Lumefantrine in Rwandese Pregnant Women Treated for Uncomplicated Plasmodium falciparum Malaria.Antimicrob Agents Chemother. 2018 Sep 24;62(10):e00518-18. doi: 10.1128/AAC.00518-18. Print 2018 Oct. Antimicrob Agents Chemother. 2018. PMID: 30061282 Free PMC article.
-
Malaria prevalence, severity and treatment outcome in relation to day 7 lumefantrine plasma concentration in pregnant women.Malar J. 2016 May 13;15(1):278. doi: 10.1186/s12936-016-1327-1. Malar J. 2016. PMID: 27177586 Free PMC article.
References
-
- Rogerson SJ, Mwapasa V, Meshnick SR. 2007. Malaria in pregnancy: linking immunity and pathogenesis to prevention. Am. J. Trop. Med. Hyg. 77(Suppl 6):14–22 - PubMed
-
- Steketee RW, Nahlen BL, Parise ME, Menendez C. 2001. The burden of malaria in pregnancy in malaria-endemic areas. Am. J. Trop. Med. Hyg. 64(Suppl 1-2):28–35 - PubMed
-
- Piola P, Nabasumba C, Turyakira E, Dhorda M, Lindegardh N, Nyehangane D, Snounou G, Ashley EA, McGready R, Nosten F, Guerin PJ. 2010. Efficacy and safety of artemether-lumefantrine compared with quinine in pregnant women with uncomplicated Plasmodium falciparum malaria: an open-label, randomised, non-inferiority trial. Lancet Infect. Dis. 10:762–769. 10.1016/S1473-3099(10)70202-4 - DOI - PubMed
-
- World Health Organization. 2010. Guidelines for the treatment of malaria, 2nd ed. World Health Organization, Geneva, Switzerland
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

