UCHL1 deficiency exacerbates human islet amyloid polypeptide toxicity in β-cells: evidence of interplay between the ubiquitin/proteasome system and autophagy
- PMID: 24879150
- PMCID: PMC4091165
- DOI: 10.4161/auto.28478
UCHL1 deficiency exacerbates human islet amyloid polypeptide toxicity in β-cells: evidence of interplay between the ubiquitin/proteasome system and autophagy
Abstract
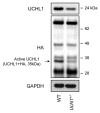
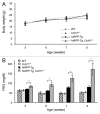
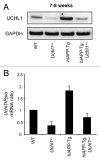
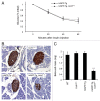
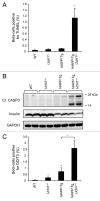
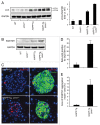
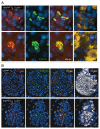
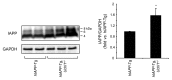
The islet in type 2 diabetes mellitus (T2DM) is characterized by a deficit in β-cells and increased β-cell apoptosis attributable at least in part to intracellular toxic oligomers of IAPP (islet amyloid polypeptide). β-cells of individuals with T2DM are also characterized by accumulation of polyubiquitinated proteins and deficiency in the deubiquitinating enzyme UCHL1 (ubiquitin carboxyl-terminal esterase L1 [ubiquitin thiolesterase]), accounting for a dysfunctional ubiquitin/proteasome system. In the present study, we used mouse genetics to elucidate in vivo whether a partial deficit in UCHL1 enhances the vulnerability of β-cells to human-IAPP (hIAPP) toxicity, and thus accelerates diabetes onset. We further investigated whether a genetically induced deficit in UCHL1 function in β-cells exacerbates hIAPP-induced alteration of the autophagy pathway in vivo. We report that a deficit in UCHL1 accelerated the onset of diabetes in hIAPP transgenic mice, due to a decrease in β-cell mass caused by increased β-cell apoptosis. We report that UCHL1 dysfunction aggravated the hIAPP-induced defect in the autophagy/lysosomal pathway, illustrated by the marked accumulation of autophagosomes and cytoplasmic inclusions positive for SQSTM1/p62 and polyubiquitinated proteins with lysine 63-specific ubiquitin chains. Collectively, this study shows that defective UCHL1 function may be an early contributor to vulnerability of pancreatic β-cells for protein misfolding and proteotoxicity, hallmark defects in islets of T2DM. Also, given that deficiency in UCHL1 exacerbated the defective autophagy/lysosomal degradation characteristic of hIAPP proteotoxicity, we demonstrate a previously unrecognized role of UCHL1 in the function of the autophagy/lysosomal pathway in β-cells.
Keywords: SQSTM1/p62; apoptosis; autophagy; diabetes; islet amyloid polypeptide; ubiquitin carboxyl-terminal esterase L1; β-cell.
Figures
Similar articles
-
β-cell dysfunctional ERAD/ubiquitin/proteasome system in type 2 diabetes mediated by islet amyloid polypeptide-induced UCH-L1 deficiency.Diabetes. 2011 Jan;60(1):227-38. doi: 10.2337/db10-0522. Epub 2010 Oct 27. Diabetes. 2011. PMID: 20980462 Free PMC article.
-
Amyloidogenic peptide oligomer accumulation in autophagy-deficient β cells induces diabetes.J Clin Invest. 2014 Aug;124(8):3311-24. doi: 10.1172/JCI69625. Epub 2014 Jul 18. J Clin Invest. 2014. PMID: 25036705 Free PMC article.
-
Human IAPP-induced pancreatic β cell toxicity and its regulation by autophagy.J Clin Invest. 2014 Aug;124(8):3634-44. doi: 10.1172/JCI69866. Epub 2014 Jul 18. J Clin Invest. 2014. PMID: 25036706 Free PMC article.
-
Human IAPP amyloidogenic properties and pancreatic β-cell death.Cell Calcium. 2014 Nov;56(5):416-27. doi: 10.1016/j.ceca.2014.08.011. Epub 2014 Aug 27. Cell Calcium. 2014. PMID: 25224501 Review.
-
The β-cell assassin: IAPP cytotoxicity.J Mol Endocrinol. 2017 Oct;59(3):R121-R140. doi: 10.1530/JME-17-0105. Epub 2017 Aug 15. J Mol Endocrinol. 2017. PMID: 28811318 Review.
Cited by
-
Phosphoproteomic profiling of selenate-treated Alzheimer's disease model cells.PLoS One. 2014 Dec 8;9(12):e113307. doi: 10.1371/journal.pone.0113307. eCollection 2014. PLoS One. 2014. PMID: 25485856 Free PMC article.
-
Bone marrow-derived mesenchymal stem cells ameliorate chronic high glucose-induced β-cell injury through modulation of autophagy.Cell Death Dis. 2015 Sep 17;6(9):e1885. doi: 10.1038/cddis.2015.230. Cell Death Dis. 2015. PMID: 26379190 Free PMC article.
-
The overexpression of human amylin in pancreatic β cells facilitate the appearance of amylin aggregates in the kidney contributing to diabetic nephropathy.Sci Rep. 2024 Oct 21;14(1):24729. doi: 10.1038/s41598-024-77063-9. Sci Rep. 2024. PMID: 39433955 Free PMC article.
-
Identification of the Shared Gene Signatures Between Alzheimer's Disease and Diabetes-Associated Cognitive Dysfunction by Bioinformatics Analysis Combined with Biological Experiment.J Alzheimers Dis. 2024;101(2):611-625. doi: 10.3233/JAD-240353. J Alzheimers Dis. 2024. PMID: 39213070 Free PMC article.
-
Hydrogen sulfide plays a potential alternative for the treatment of metabolic disorders of diabetic cardiomyopathy.Mol Cell Biochem. 2022 Jan;477(1):255-265. doi: 10.1007/s11010-021-04278-z. Epub 2021 Oct 23. Mol Cell Biochem. 2022. PMID: 34687394 Review.
References
-
- Gurlo T, Ryazantsev S, Huang CJ, Yeh MW, Reber HA, Hines OJ, O’Brien TD, Glabe CG, Butler PC. Evidence for proteotoxicity in beta cells in type 2 diabetes: toxic islet amyloid polypeptide oligomers form intracellularly in the secretory pathway. Am J Pathol. 2010;176:861–9. doi: 10.2353/ajpath.2010.090532. - DOI - PMC - PubMed
-
- Huang CJ, Haataja L, Gurlo T, Butler AE, Wu X, Soeller WC, Butler PC. Induction of endoplasmic reticulum stress-induced beta-cell apoptosis and accumulation of polyubiquitinated proteins by human islet amyloid polypeptide. Am J Physiol Endocrinol Metab. 2007;293:E1656–62. doi: 10.1152/ajpendo.00318.2007. - DOI - PubMed
-
- Huang CJ, Lin CY, Haataja L, Gurlo T, Butler AE, Rizza RA, Butler PC. High expression rates of human islet amyloid polypeptide induce endoplasmic reticulum stress mediated beta-cell apoptosis, a characteristic of humans with type 2 but not type 1 diabetes. Diabetes. 2007;56:2016–27. doi: 10.2337/db07-0197. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
