Phosphorylation of NBR1 by GSK3 modulates protein aggregation
- PMID: 24879152
- PMCID: PMC4091167
- DOI: 10.4161/auto.28479
Phosphorylation of NBR1 by GSK3 modulates protein aggregation
Abstract
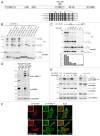
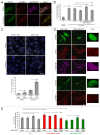
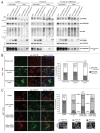
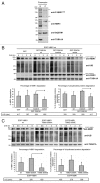
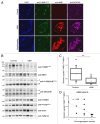
The autophagy receptor NBR1 (neighbor of BRCA1 gene 1) binds UB/ubiquitin and the autophagosome-conjugated MAP1LC3/LC3 (microtubule-associated protein 1 light chain 3) proteins, thereby ensuring ubiquitinated protein degradation. Numerous neurodegenerative and neuromuscular diseases are associated with inappropriate aggregation of ubiquitinated proteins and GSK3 (glycogen synthase kinase 3) activity is involved in several of these proteinopathies. Here we show that NBR1 is a substrate of GSK3. NBR1 phosphorylation by GSK3 at Thr586 prevents the aggregation of ubiquitinated proteins and their selective autophagic degradation. Indeed, NBR1 phosphorylation decreases protein aggregation induced by puromycin or by the DES/desmin N342D mutant found in desminopathy patients and stabilizes ubiquitinated proteins. Importantly, decrease of protein aggregates is due to an inhibition of their formation and not to their autophagic degradation as confirmed by data on Atg7 knockout mice. The relevance of NBR1 phosphorylation in human pathology was investigated. Analysis of muscle biopsies of sporadic inclusion body myositis (sIBM) patients revealed a strong decrease of NBR1 phosphorylation in muscles of sIBM patients that directly correlated with the severity of protein aggregation. We propose that phosphorylation of NBR1 by GSK3 modulates the formation of protein aggregates and that this regulation mechanism is defective in a human muscle proteinopathy.
Keywords: GSK3; NBR1; autophagy; sIBM; skeletal muscle; sporadic inclusion body myositis; ubiquitinated protein aggregates.
Figures
Similar articles
-
Abnormalities of NBR1, a novel autophagy-associated protein, in muscle fibers of sporadic inclusion-body myositis.Acta Neuropathol. 2011 Nov;122(5):627-36. doi: 10.1007/s00401-011-0874-3. Epub 2011 Sep 21. Acta Neuropathol. 2011. PMID: 21935636
-
A role for NBR1 in autophagosomal degradation of ubiquitinated substrates.Mol Cell. 2009 Feb 27;33(4):505-16. doi: 10.1016/j.molcel.2009.01.020. Mol Cell. 2009. PMID: 19250911
-
NBR1 cooperates with p62 in selective autophagy of ubiquitinated targets.Autophagy. 2009 Jul;5(5):732-3. doi: 10.4161/auto.5.5.8566. Epub 2009 Jul 30. Autophagy. 2009. PMID: 19398892
-
Broad and Complex Roles of NBR1-Mediated Selective Autophagy in Plant Stress Responses.Cells. 2020 Nov 30;9(12):2562. doi: 10.3390/cells9122562. Cells. 2020. PMID: 33266087 Free PMC article. Review.
-
Acetylation in the regulation of autophagy.Autophagy. 2023 Feb;19(2):379-387. doi: 10.1080/15548627.2022.2062112. Epub 2022 Apr 18. Autophagy. 2023. PMID: 35435793 Free PMC article. Review.
Cited by
-
NBR1: The archetypal selective autophagy receptor.J Cell Biol. 2022 Nov 7;221(11):e202208092. doi: 10.1083/jcb.202208092. Epub 2022 Oct 18. J Cell Biol. 2022. PMID: 36255390 Free PMC article. Review.
-
Mitochondrial defects in sporadic inclusion body myositis-causes and consequences.Front Cell Dev Biol. 2024 May 14;12:1403463. doi: 10.3389/fcell.2024.1403463. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 38808223 Free PMC article. Review.
-
Mitochondrial Quality Control and Muscle Mass Maintenance.Front Physiol. 2016 Jan 12;6:422. doi: 10.3389/fphys.2015.00422. eCollection 2015. Front Physiol. 2016. PMID: 26793123 Free PMC article. Review.
-
New insights into the protein aggregation pathology in myotilinopathy by combined proteomic and immunolocalization analyses.Acta Neuropathol Commun. 2016 Feb 3;4:8. doi: 10.1186/s40478-016-0280-0. Acta Neuropathol Commun. 2016. PMID: 26842778 Free PMC article.
-
Hypoxia signaling pathways: modulators of oxygen-related organelles.Front Cell Dev Biol. 2015 Jul 21;3:42. doi: 10.3389/fcell.2015.00042. eCollection 2015. Front Cell Dev Biol. 2015. PMID: 26258123 Free PMC article. Review.
References
-
- Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, Scaravilli F, Easton DF, Duden R, O’Kane CJ, et al. Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet. 2004;36:585–95. doi: 10.1038/ng1362. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
