RNA polymerase II transcription elongation and Pol II CTD Ser2 phosphorylation: A tail of two kinases
- PMID: 24879308
- PMCID: PMC4133218
- DOI: 10.4161/nucl.29347
RNA polymerase II transcription elongation and Pol II CTD Ser2 phosphorylation: A tail of two kinases
Abstract
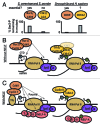
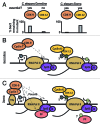
The transition between initiation and productive elongation during RNA Polymerase II (Pol II) transcription is a well-appreciated point of regulation across many eukaryotes. Elongating Pol II is modified by phosphorylation of serine 2 (Ser2) on its carboxy terminal domain (CTD) by two kinases, Bur1/Ctk1 in yeast and Cdk9/Cdk12 in metazoans. Here, we discuss the roles and regulation of these kinases and their relationship to Pol II elongation control, and focus on recent data from work in C. elegans that point out gaps in our current understand of transcription elongation.
Keywords: Bur1; C. elegans; Cdk12; Cdk9; Ctk1; P-TEFb; RNA Polymerase II; Serine 2; elongation; transcription.
Figures
References
-
- Cadena DL, Dahmus ME. Messenger RNA synthesis in mammalian cells is catalyzed by the phosphorylated form of RNA polymerase II. J Biol Chem. 1987;262:12468–74. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
