A highly processive topoisomerase I: studies at the single-molecule level
- PMID: 24880688
- PMCID: PMC4081095
- DOI: 10.1093/nar/gku494
A highly processive topoisomerase I: studies at the single-molecule level
Abstract
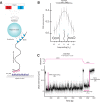
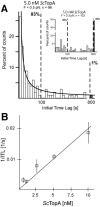
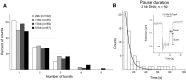
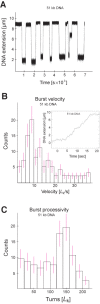
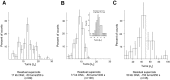
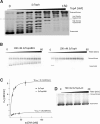
Amongst enzymes which relieve torsional strain and maintain chromosome supercoiling, type IA topoisomerases share a strand-passage mechanism that involves transient nicking and re-joining of a single deoxyribonucleic acid (DNA) strand. In contrast to many bacterial species that possess two type IA topoisomerases (TopA and TopB), Actinobacteria possess only TopA, and unlike its homologues this topoisomerase has a unique C-terminal domain that lacks the Zn-finger motifs characteristic of type IA enzymes. To better understand how this unique C-terminal domain affects the enzyme's activity, we have examined DNA relaxation by actinobacterial TopA from Streptomyces coelicolor (ScTopA) using real-time single-molecule experiments. These studies reveal extremely high processivity of ScTopA not described previously for any other topoisomerase of type I. Moreover, we also demonstrate that enzyme processivity varies in a torque-dependent manner. Based on the analysis of the C-terminally truncated ScTopA mutants, we propose that high processivity of the enzyme is associated with the presence of a stretch of positively charged amino acids in its C-terminal region.
© The Author(s) 2014. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures

Similar articles
-
A highly processive actinobacterial topoisomerase I - thoughts on Streptomyces' demand for an enzyme with a unique C-terminal domain.Microbiology (Reading). 2020 Feb;166(2):120-128. doi: 10.1099/mic.0.000841. Epub 2019 Aug 7. Microbiology (Reading). 2020. PMID: 31390324 Free PMC article. Review.
-
C-terminal lysine repeats in Streptomyces topoisomerase I stabilize the enzyme-DNA complex and confer high enzyme processivity.Nucleic Acids Res. 2017 Nov 16;45(20):11908-11924. doi: 10.1093/nar/gkx827. Nucleic Acids Res. 2017. PMID: 28981718 Free PMC article.
-
The Coordinated Positive Regulation of Topoisomerase Genes Maintains Topological Homeostasis in Streptomyces coelicolor.J Bacteriol. 2016 Oct 7;198(21):3016-3028. doi: 10.1128/JB.00530-16. Print 2016 Nov 1. J Bacteriol. 2016. PMID: 27551021 Free PMC article.
-
Friction and torque govern the relaxation of DNA supercoils by eukaryotic topoisomerase IB.Nature. 2005 Mar 31;434(7033):671-4. doi: 10.1038/nature03395. Nature. 2005. PMID: 15800630
-
[DNA supercoiling and topoisomerases in Escherichia coli].Rev Latinoam Microbiol. 1995 Jul-Sep;37(3):291-304. Rev Latinoam Microbiol. 1995. PMID: 8850348 Review. Spanish.
Cited by
-
Mechanism of Type IA Topoisomerases.Molecules. 2020 Oct 17;25(20):4769. doi: 10.3390/molecules25204769. Molecules. 2020. PMID: 33080770 Free PMC article. Review.
-
Amsacrine Derivatives Selectively Inhibit Mycobacterial Topoisomerase I (TopA), Impair M. smegmatis Growth and Disturb Chromosome Replication.Front Microbiol. 2018 Jul 17;9:1592. doi: 10.3389/fmicb.2018.01592. eCollection 2018. Front Microbiol. 2018. PMID: 30065714 Free PMC article.
-
Structural basis for suppression of hypernegative DNA supercoiling by E. coli topoisomerase I.Nucleic Acids Res. 2015 Dec 15;43(22):11031-46. doi: 10.1093/nar/gkv1073. Epub 2015 Oct 20. Nucleic Acids Res. 2015. PMID: 26490962 Free PMC article.
-
Targeting bacterial topoisomerase I to meet the challenge of finding new antibiotics.Future Med Chem. 2015;7(4):459-71. doi: 10.4155/fmc.14.157. Future Med Chem. 2015. PMID: 25875873 Free PMC article. Review.
-
Transcriptional Response of Streptomyces coelicolor to Rapid Chromosome Relaxation or Long-Term Supercoiling Imbalance.Front Microbiol. 2019 Jul 11;10:1605. doi: 10.3389/fmicb.2019.01605. eCollection 2019. Front Microbiol. 2019. PMID: 31354687 Free PMC article.
References
-
- Everid A.C., Small J.V., Davies H.G. Electron-microscope observations on the structure of condensed chromatin: evidence for orderly arrays of unit threads on the surface of chicken erythrocyte nuclei. J. Cell Sci. 1970;7:35–48. - PubMed
-
- Richmond T.J., Davey C.A. The structure of DNA in the nucleosome core. Nature. 2003;423:145–150. - PubMed
-
- Zimmerman S.B. Cooperative transitions of isolated Escherichia coli nucleoids: implications for the nucleoid as a cellular phase. J. Struct. Biol. 2006;153:160–175. - PubMed
-
- Zimmerman S.B. Shape and compaction of Escherichia coli nucleoids. J. Struct. Biol. 2006;156:255–261. - PubMed
-
- Nasmyth K., Haering C.H. The structure and function of SMC and kleisin complexes. Annu. Rev. Biochem. 2005;74:595–648. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

