Characterization of murine pituitary-derived cell lines Tpit/F1, Tpit/E and TtT/GF
- PMID: 24881870
- PMCID: PMC4139504
- DOI: 10.1262/jrd.2014-031
Characterization of murine pituitary-derived cell lines Tpit/F1, Tpit/E and TtT/GF
Abstract
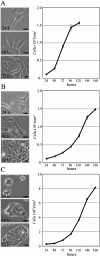
The pituitary is an important endocrine tissue of the vertebrate that produces and secretes many hormones. Accumulating data suggest that several types of cells compose the pituitary, and there is growing interest in elucidating the origin of these cell types and their roles in pituitary organogenesis. Therein, the histogenous cell line is an extremely valuable experimental tool for investigating the function of derived tissue. In this study, we compared gene expression profiles by microarray analysis and real-time PCR for murine pituitary tumor-derived non-hormone-producing cell lines TtT/GF, Tpit/F1 and Tpit/E. Several genes are characteristically expressed in each cell line: Abcg2, Nestin, Prrx1, Prrx2, CD34, Eng, Cspg4 (Ng2), S100β and nNos in TtT/GF; Cxcl12, Raldh1, Msx1 and Twist1 in Tpit/F1; and Cxadr, Sox9, Cdh1, EpCAM and Krt8 in Tpit/E. Ultimately, we came to the following conclusions: TtT/GF cells show the most differentiated state, and may have some properties of the pituitary vascular endothelial cell and/or pericyte. Tpit/F1 cells show the epithelial and mesenchymal phenotypes with stemness still in a transiting state. Tpit/E cells have a phenotype of epithelial cells and are the most immature cells in the progression of differentiation or in the initial endothelial-mesenchymal transition (EMT). Thus, these three cell lines must be useful model cell lines for investigating pituitary stem/progenitor cells as well as organogenesis.
Figures


Similar articles
-
Clump formation in mouse pituitary-derived non-endocrine cell line Tpit/F1 promotes differentiation into growth-hormone-producing cells.Cell Tissue Res. 2017 Aug;369(2):353-368. doi: 10.1007/s00441-017-2603-2. Epub 2017 Mar 31. Cell Tissue Res. 2017. PMID: 28364143
-
Characterization of a pituitary-tumor-derived cell line, TtT/GF, that expresses Hoechst efflux ABC transporter subfamily G2 and stem cell antigen 1.Cell Tissue Res. 2013 Nov;354(2):563-72. doi: 10.1007/s00441-013-1686-7. Epub 2013 Jul 24. Cell Tissue Res. 2013. PMID: 23881407
-
TGFβ signaling reinforces pericyte properties of the non-endocrine mouse pituitary cell line TtT/GF.Cell Tissue Res. 2018 Feb;371(2):339-350. doi: 10.1007/s00441-017-2758-x. Epub 2017 Dec 22. Cell Tissue Res. 2018. PMID: 29274061
-
[Tpit mutations reveal a new model of pituitary differentiation and account for isolated ACTH deficiency].Med Sci (Paris). 2004 Nov;20(11):1009-13. doi: 10.1051/medsci/200420111009. Med Sci (Paris). 2004. PMID: 15525497 Review. French.
-
Pituitary cell lines and their endocrine applications.Mol Cell Endocrinol. 2004 Dec 30;228(1-2):1-21. doi: 10.1016/j.mce.2004.07.018. Mol Cell Endocrinol. 2004. PMID: 15541569 Review.
Cited by
-
Involvement of GPR4 in increased growth hormone and prolactin expressions by extracellular acidification in MtT/S cells.J Reprod Dev. 2020 Apr 10;66(2):175-180. doi: 10.1262/jrd.2019-159. Epub 2020 Jan 19. J Reprod Dev. 2020. PMID: 31956173 Free PMC article.
-
New insights into the role and origin of pituitary S100β-positive cells.Cell Tissue Res. 2021 Nov;386(2):227-237. doi: 10.1007/s00441-021-03523-7. Epub 2021 Sep 22. Cell Tissue Res. 2021. PMID: 34550453 Review.
References
-
- Zhu X, Gleiberman AS, Rosenfeld MG. Molecular physiology of pituitary development: signaling and transcriptional networks. Physiol Rev 2007; 87: 933–963 - PubMed
-
- Watkins-Chow DE, Camper SA. How many homeobox genes does it take to make a pituitary gland? Trends Genet 1998; 14: 284–290 - PubMed
-
- Zhu X, Rosenfeld MG. Transcriptional control of precursor proliferation in the early phases of pituitary development. Curr Opin Genet Dev 2004; 14: 567–574 - PubMed
-
- Davis SW, Castinetti F, Carvalho LR, Ellsworth BS, Potok MA, Lyons RH, Brinkmeier ML, Raetzman LT, Carninci P, Mortensen AH, Hayashizaki Y, Arnhold IJ, Mendonça BB, Brue T, Camper SA. Molecular mechanisms of pituitary organogenesis: In search of novel regulatory genes. Mol Cell Endocrinol 2010; 323: 4–19 - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

