Direct autofluorescence during CO2 laser surgery of the larynx: can it really help the surgeon?
- PMID: 24882926
- PMCID: PMC4035836
Direct autofluorescence during CO2 laser surgery of the larynx: can it really help the surgeon?
Abstract
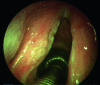
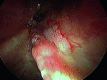
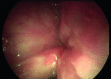
Herein we assessed the impact of direct autofluorescence during intraoperative work-up on obtaining superficial free resection margins, identifying new areas of malignant transformation and altering disease-free survival and local control at 3 years in patients submitted to transoral laser surgery (TLS) for early glottic cancer. Prospective cohort evaluation was carried out on the diagnostic accuracy of the superficial extent and TNM staging in 73 patients with glottic carcinoma undergoing transoral CO2 laser surgery. The use of direct autofluorescence was associated with superficial disease-free margins in 97.2% of cases, and with superficial close margins in 2.8%. The improvement in diagnostic accuracy was 16.4%; in 8.2% of cases, there was upstaging of the TNM classification (in one case, a second neoplastic area in a different laryngeal site was observed and considered to be a second endolaryngeal primary). The sensitivity of direct autofluorescence was 96.5% with a specificity of 98.5%. Overall, 3-year disease-specific survival and local control with laser alone were, respectively: T1a (97.5%, 100%), T1b (86.7%, 86.7%), T2 (88.9%, 88.9%). This study demonstrates that direct autofluorescence can help to identify positive superficial margins, and has a favourable impact on disease-specific survival and local control at 3 years.
Obiettivo di questo studio è stato valutare l'impatto dell'autofluorescenza diretta durante il work-up intraoperatorio nell'ottenere margini di resezione superficiale indenni, nell'individuare nuove aree di trasformazione maligna e nel migliorare a 3 anni la sopravvivenza libera da malattia/controllo locale nei pazienti sottoposti a laserchirurgia transorale (TLS) per cancro della glottide in fase iniziale. (Disegno dello studio: studio di coorte prospettico). Una valutazione prospettica sull'accuratezza diagnostica dell'estensione superficiale e sulla stadiazione TNM è stata condotta in 73 pazienti con carcinoma della glottide sottoposti a laserchirurgia transorale. L'utilizzo dell'autofluorescenza diretta ha determinato margini superficiali liberi da malattia nel 97,2 % dei casi e margini superficiali esigui nel 2,8 %. Un miglioramento nell'accuratezza diagnostica si è verificato nel 16,4 % mentre nell'8,2 % dei casi si è assistito ad un up-staging della classificazione TNM (in un caso un'area neoplastica in una sede laringea distinta è stata considerata un secondo tumore endolaringeo primitivo). La sensibilità dell'autofluorescenza diretta è stata del 96,5%, la specificità del 98,5%. Nel complesso la sopravvivenza malattia-specifica ed il controllo locale con laser a 3 anni sono stati rispettivamente: T1a (97,5 % ,100 %), T1b (86,7%, 86,7%), T2 (88,9%, 88,9%). Concludendo questo studio dimostra che l'autofluorescenza diretta può aiutare il chirurgo nell'identificare i margini superficiali positivi e risulta associata ad un impatto favorevole sulla sopravvivenza malattia-specifica e sul controllo locale a 3 anni.
Keywords: Autofluorescence; Endoscopic surgery; Glottic tumour; Laryngeal cancer; Resection margins; Transoral laser surgery.
Figures





References
-
- Peretti G, Piazza C, Cocco D, et al. Transoral CO2 laser treatment for Tis-T3 glottic cancer: the University of Brescia experience on 595 patients. Head Neck. 2010;32:977–983. - PubMed
-
- Steiner W. Experience in endoscopic laser surgery of malignant tumors of the upper aero-digestive tract. Adv Otorhinolaryngol. 1988;39:135–144. - PubMed
-
- Lucioni M, Bertolin A, D'Ascanio L, et al. Margin photocoagulation in laser surgery for early glottic cancer: impact on disease local control. Otolaryngol Head Neck Surg. 2012;146:600–605. - PubMed
-
- Steiner W, Ambrosch P. Endoscopic laser surgery of the upper aerodigestive tract. Stuttgart: Thieme; 2000. pp. 34–36.
-
- Ambrosch P. The role of laser microsurgery in the treatment of laryngeal cancer. Curr Opin Otolaryngol Head Neck Surg. 2007;15:82–88. - PubMed
