Grape seed extract prevents skeletal muscle wasting in interleukin 10 knockout mice
- PMID: 24884473
- PMCID: PMC4041050
- DOI: 10.1186/1472-6882-14-162
Grape seed extract prevents skeletal muscle wasting in interleukin 10 knockout mice
Abstract
Background: Muscle wasting is frequently a result of cancers, AIDS, chronic diseases and aging, which often links to muscle inflammation. Although grape seed extract (GSE) has been widely used as a human dietary supplement for health promotion and disease prevention primarily due to its anti-oxidative and anti-inflammative effects, it is unknown whether GSE affects muscle wasting. The objective is to test the effects of GSE supplementation on inflammation and muscle wasting in interleukin (IL)-10 knockout mice, a recently developed model for human frailty.
Methods: Male IL-10 knockout (IL10KO) C57BL/6 mice at 6 weeks of age were assigned to either 0% or 0.1% GSE (in drinking water) groups (n=10) for 12 weeks, when skeletal muscle was sampled for analyses. Wild-type C57BL/6 male mice were used as controls.
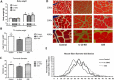
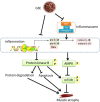
Results: Tibialis anterior muscle weight and fiber size of IL10KO mice were much lower than wild-type mice. IL10KO enhanced nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling and inflammasome formation when compared to wild-type mice. Phosphorylation of anabolic signaling was inhibited, whereas muscle specific ubiquitin ligase, AMP-activated protein kinase (AMPK) and apoptotic signaling were up-regulated in IL10KO mice. GSE supplementation effectively rectified these adverse changes in IL10KO muscle, which provide an explanation for the enhanced muscle mass, reduced protein degradation and apoptosis in GSE supplemented mice compared to IL10KO mice without supplementation.
Conclusion: GSE supplementation effectively prevents muscle wasting in IL10KO mice, showing that GSE can be used as an auxiliary treatment for muscle loss associated with chronic inflammation and frailty.
Figures




Similar articles
-
Grape seed extract improves epithelial structure and suppresses inflammation in ileum of IL-10-deficient mice.Food Funct. 2014 Oct;5(10):2558-63. doi: 10.1039/c4fo00451e. Epub 2014 Aug 19. Food Funct. 2014. PMID: 25137131
-
Grape seed extract efficacy against azoxymethane-induced colon tumorigenesis in A/J mice: interlinking miRNA with cytokine signaling and inflammation.Cancer Prev Res (Phila). 2013 Jul;6(7):625-33. doi: 10.1158/1940-6207.CAPR-13-0053. Epub 2013 May 2. Cancer Prev Res (Phila). 2013. PMID: 23639480 Free PMC article.
-
Dietary grape seed extract ameliorates symptoms of inflammatory bowel disease in IL10-deficient mice.Mol Nutr Food Res. 2013 Dec;57(12):2253-7. doi: 10.1002/mnfr.201300146. Epub 2013 Aug 21. Mol Nutr Food Res. 2013. PMID: 23963706 Free PMC article.
-
Triggers and mechanisms of skeletal muscle wasting in chronic obstructive pulmonary disease.Int J Biochem Cell Biol. 2013 Oct;45(10):2245-56. doi: 10.1016/j.biocel.2013.06.015. Epub 2013 Jul 1. Int J Biochem Cell Biol. 2013. PMID: 23827718 Review.
-
Nuclear factor-kappa B signaling in skeletal muscle atrophy.J Mol Med (Berl). 2008 Oct;86(10):1113-26. doi: 10.1007/s00109-008-0373-8. Epub 2008 Jun 24. J Mol Med (Berl). 2008. PMID: 18574572 Free PMC article. Review.
Cited by
-
Retinoic acid induces white adipose tissue browning by increasing adipose vascularity and inducing beige adipogenesis of PDGFRα+ adipose progenitors.Cell Discov. 2017 Oct 10;3:17036. doi: 10.1038/celldisc.2017.36. eCollection 2017. Cell Discov. 2017. PMID: 29021914 Free PMC article.
-
Role of Inflammation in Muscle Homeostasis and Myogenesis.Mediators Inflamm. 2015;2015:805172. doi: 10.1155/2015/805172. Epub 2015 Oct 5. Mediators Inflamm. 2015. PMID: 26508819 Free PMC article. Review.
-
Polyphenol-Enriched Plum Extract Enhances Myotubule Formation and Anabolism while Attenuating Colon Cancer-induced Cellular Damage in C2C12 Cells.Nutrients. 2019 May 15;11(5):1077. doi: 10.3390/nu11051077. Nutrients. 2019. PMID: 31096595 Free PMC article.
-
Moderate alcohol intake induces thermogenic brown/beige adipocyte formation via elevating retinoic acid signaling.FASEB J. 2017 Oct;31(10):4612-4622. doi: 10.1096/fj.201700396R. Epub 2017 Jul 5. FASEB J. 2017. PMID: 28679528 Free PMC article.
-
Inflammatory Markers Associated with Physical Frailty and Cognitive Impairment.Aging Dis. 2024 Apr 17;16(2):859-875. doi: 10.14336/AD.2024.0258. Aging Dis. 2024. PMID: 38739942 Free PMC article. Review.
References
-
- Mosoni L, Malmezat T, Valluy MC, Houlier ML, Attaix D, Mirand PP. Lower recovery of muscle protein lost during starvation in old rats despite a stimulation of protein synthesis. Am J Physiol. 1999;277(4 Pt 1):E608–E616. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

