TIMP-1 and responsiveness to gemcitabine in advanced breast cancer; results from a randomized phase III trial from the Danish breast cancer cooperative group
- PMID: 24884504
- PMCID: PMC4091674
- DOI: 10.1186/1471-2407-14-360
TIMP-1 and responsiveness to gemcitabine in advanced breast cancer; results from a randomized phase III trial from the Danish breast cancer cooperative group
Abstract
Background: Tissue inhibitor of metalloproteinases-1 (TIMP-1) has anti-apoptotic functions, which may protect TIMP-1 positive cancer cells from the effects of chemotherapy such as docetaxel and gemcitabine. The purpose of the present study was to evaluate TIMP-1 immunoreactivity as a prognostic and predictive marker in advanced breast cancer patients receiving docetaxel (D) or gemcitabine plus docetaxel (GD).
Methods: Patients with locally advanced or metastatic breast cancer who were assigned to D or GD by participation in a randomized phase III trial were included in the study. Assessment of TIMP-1 status was performed retrospectively on primary tumor whole-tissue sections by immunohistochemistry and tumor samples were considered positive if epithelial breast cancer cells were stained by the anti-TIMP-1 monoclonal antibody VT7. Time to progression (TTP) was the primary endpoint. Overall survival (OS) and response rate (RR) were secondary endpoints. Associations between TIMP-1 status and outcome after chemotherapy were analyzed by Kaplan-Meier estimates and Cox proportional hazards regression models.
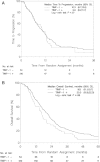
Results: TIMP-1 status was available from 264 of 337 patients and 210 (80%) of the tumors were classified as cancer cell TIMP-1 positive. No significant difference for TTP between TIMP-1 positive versus TIMP-1 negative patients was observed in multivariate analysis, and RR did not differ according to TIMP-1 status. However, patients with TIMP-1 positive tumors had a significant reduction in OS events (hazard ratio = 0.71, 95% confidence interval (CI) = 0.52-0.98, P = 0.03). Additionally, a borderline significant interaction for OS was observed between TIMP-1 status and benefit from GD compared to D (Pinteraction = 0.06) such that median OS increased by nine months for TIMP-1 negative patients receiving GD.
Conclusions: TIMP-1 status was an independent prognostic factor for OS but not TTP in patients with advanced breast cancer receiving either D or GD. There was no statistically significant interaction between TIMP-1 status and treatment, but a trend towards an incremental OS from the addition of gemcitabine to docetaxel in patients with TIMP-1 negative tumors suggests further investigation.
Figures


Similar articles
-
Gemcitabine plus docetaxel versus docetaxel in patients with predominantly human epidermal growth factor receptor 2-negative locally advanced or metastatic breast cancer: a randomized, phase III study by the Danish Breast Cancer Cooperative Group.J Clin Oncol. 2011 Dec 20;29(36):4748-54. doi: 10.1200/JCO.2010.33.9507. Epub 2011 Nov 14. J Clin Oncol. 2011. PMID: 22084374 Clinical Trial.
-
PAM50 breast cancer intrinsic subtypes and effect of gemcitabine in advanced breast cancer patients.Acta Oncol. 2014 Jun;53(6):776-87. doi: 10.3109/0284186X.2013.865076. Epub 2013 Dec 20. Acta Oncol. 2014. PMID: 24359601
-
A pooled analysis of gemcitabine plus docetaxel versus capecitabine plus docetaxel in metastatic breast cancer.Oncologist. 2014 May;19(5):443-52. doi: 10.1634/theoncologist.2013-0428. Epub 2014 Apr 4. Oncologist. 2014. PMID: 24705980 Free PMC article. Clinical Trial.
-
Role of gemcitabine in metastatic breast cancer patients: a short review.Breast. 2008 Jun;17(3):220-6. doi: 10.1016/j.breast.2007.10.009. Epub 2007 Nov 26. Breast. 2008. PMID: 18037292 Review.
-
Overview of gemcitabine activity in advanced breast cancer.Semin Oncol. 2006 Jun;33(3 Suppl 9):S19-23. doi: 10.1053/j.seminoncol.2006.03.020. Semin Oncol. 2006. PMID: 16797378 Review.
References
-
- Ejlertsen B, Jensen MB, Nielsen KV, Balslev E, Rasmussen BB, Willemoe GL, Hertel PB, Knoop AS, Mouridsen HT, Brunner N. HER2, TOP2A, and TIMP-1 and responsiveness to adjuvant anthracycline-containing chemotherapy in high-risk breast cancer patients. J Clin Oncol. 2010;28:984–990. - PubMed
-
- Henriksen KL, Rasmussen BB, Lykkesfeldt AE, Moller S, Ejlertsen B, Mouridsen HT. An ER activity profile including ER, PR, Bcl-2 and IGF-IR may have potential as selection criterion for letrozole or tamoxifen treatment of patients with advanced breast cancer. Acta Oncol. 2009;48:522–531. - PubMed
-
- Knoop AS, Knudsen H, Balslev E, Rasmussen BB, Overgaard J, Nielsen KV, Schonau A, Gunnarsdottir K, Olsen KE, Mouridsen H, Ejlertsen B, Danish Breast Cancer Cooperative G. Retrospective analysis of topoisomerase IIa amplifications and deletions as predictive markers in primary breast cancer patients randomly assigned to cyclophosphamide, methotrexate, and fluorouracil or cyclophosphamide, epirubicin, and fluorouracil: Danish Breast Cancer Cooperative Group. J Clin Oncol. 2005;23:7483–7490. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

