Nerve growth factor improves functional recovery by inhibiting endoplasmic reticulum stress-induced neuronal apoptosis in rats with spinal cord injury
- PMID: 24884850
- PMCID: PMC4039547
- DOI: 10.1186/1479-5876-12-130
Nerve growth factor improves functional recovery by inhibiting endoplasmic reticulum stress-induced neuronal apoptosis in rats with spinal cord injury
Erratum in
-
Correction to: Nerve growth factor improves functional recovery by inhibiting endoplasmic reticulum stress-induced neuronal apoptosis in rats with spinal cord injury.J Transl Med. 2021 Oct 25;19(1):445. doi: 10.1186/s12967-021-02901-7. J Transl Med. 2021. PMID: 34696770 Free PMC article. No abstract available.
Abstract
Background: Endoplasmic reticulum (ER) stress-induced apoptosis plays a major role in various diseases, including spinal cord injury (SCI). Nerve growth factor (NGF) show neuroprotective effect and improve the recovery of SCI, but the relations of ER stress-induced apoptosis and the NGF therapeutic effect in SCI still unclear.
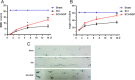
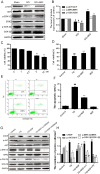
Methods: Young adult female Sprague-Dawley rats's vertebral column was exposed and a laminectomy was done at T9 vertebrae and moderate contusion injuries were performed using a vascular clip. NGF stock solution was diluted with 0.9% NaCl and administered intravenously at a dose of 20 μg/kg/day after SCI and then once per day until they were executed. Subsequently, the rats were executed at 1d, 3 d, 7d and 14d. The locomotor activities of SCI model rats were tested by the 21-point Basso-Beattie-Bresnahan (BBB) locomotion scale, inclined plane test and footprint analysis. In addition, Western blot analysis was performed to identify the expression of ER-stress related proteins including CHOP, GRP78 and caspase-12 both in vivo and in vitro. The level of cell apoptosis was determined by TUNEL in vivo and Flow cytometry in vitro. Relative downstream signals Akt/GSK-3β and ERK1/2were also analyzed with or without inhibitors in vitro.
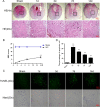
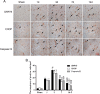
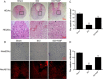
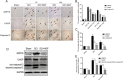
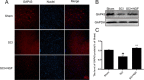
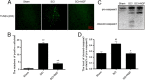
Results: Our results demonstrated that ER stress-induced apoptosis was involved in the injury of SCI model rats. NGF administration improved the motor function recovery and increased the neurons survival in the spinal cord lesions of the model rats. NGF decreases neuron apoptosis which measured by TUNEL and inhibits the activation of caspase-3 cascade. The ER stress-induced apoptosis response proteins CHOP, GRP78 and caspase-12 are inhibited by NGF treatment. Meanwhile, NGF administration also increased expression of growth-associated protein 43 (GAP43). The administration of NGF activated downstream signals Akt/GSK-3β and ERK1/2 in ER stress cell model in vitro.
Conclusion: The neuroprotective role of NGF in the recovery of SCI is related to the inhibition of ER stress-induced cell death via the activation of downstream signals, also suggested a new trend of NGF translational drug development in the central neural system injuries which involved in the regulation of chronic ER stress.
Figures

Similar articles
-
Gelatin Nanostructured Lipid Carriers Incorporating Nerve Growth Factor Inhibit Endoplasmic Reticulum Stress-Induced Apoptosis and Improve Recovery in Spinal Cord Injury.Mol Neurobiol. 2016 Sep;53(7):4375-86. doi: 10.1007/s12035-015-9372-2. Epub 2015 Aug 2. Mol Neurobiol. 2016. PMID: 26232067
-
Exogenous basic fibroblast growth factor inhibits ER stress-induced apoptosis and improves recovery from spinal cord injury.CNS Neurosci Ther. 2013 Jan;19(1):20-9. doi: 10.1111/cns.12013. Epub 2012 Oct 20. CNS Neurosci Ther. 2013. PMID: 23082997 Free PMC article.
-
Dl-3-n-butylphthalide improves functional recovery in rats with spinal cord injury by inhibiting endoplasmic reticulum stress-induced apoptosis.Am J Transl Res. 2017 Mar 15;9(3):1075-1087. eCollection 2017. Am J Transl Res. 2017. PMID: 28386335 Free PMC article.
-
Mechanism and prospects of mitochondrial transplantation for spinal cord injury treatment.Stem Cell Res Ther. 2024 Nov 28;15(1):457. doi: 10.1186/s13287-024-04077-5. Stem Cell Res Ther. 2024. PMID: 39609871 Free PMC article. Review.
-
Granulocyte colony-stimulating factor effects on neurological and motor function in animals with spinal cord injury: a systematic review and meta-analysis.Front Neurosci. 2023 Jun 28;17:1168764. doi: 10.3389/fnins.2023.1168764. eCollection 2023. Front Neurosci. 2023. PMID: 37449274 Free PMC article. Review.
Cited by
-
Effects of combination treatment with transcranial magnetic stimulation and bone marrow mesenchymal stem cell transplantation or Raf inhibition on spinal cord injury in rats.Mol Med Rep. 2021 Apr;23(4):294. doi: 10.3892/mmr.2021.11934. Epub 2021 Mar 2. Mol Med Rep. 2021. PMID: 33649786 Free PMC article.
-
The Proteostasis Network: A Global Therapeutic Target for Neuroprotection after Spinal Cord Injury.Cells. 2022 Oct 22;11(21):3339. doi: 10.3390/cells11213339. Cells. 2022. PMID: 36359735 Free PMC article. Review.
-
Sustained delivery of neurotrophic factors to treat spinal cord injury.Transl Neurosci. 2021 Nov 30;12(1):494-511. doi: 10.1515/tnsci-2020-0200. eCollection 2021 Jan 1. Transl Neurosci. 2021. PMID: 34900347 Free PMC article. Review.
-
The emerging role of cuproptosis in spinal cord injury.Front Immunol. 2025 Jun 16;16:1595852. doi: 10.3389/fimmu.2025.1595852. eCollection 2025. Front Immunol. 2025. PMID: 40589743 Free PMC article. Review.
-
Transplantation of mesenchymal stem cells promotes the functional recovery of the central nervous system following cerebral ischemia by inhibiting myelin-associated inhibitor expression and neural apoptosis.Exp Ther Med. 2016 May;11(5):1595-1600. doi: 10.3892/etm.2016.3089. Epub 2016 Feb 19. Exp Ther Med. 2016. PMID: 27168778 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

