Singlet oxygen generation on porous superhydrophobic surfaces: effect of gas flow and sensitizer wetting on trapping efficiency
- PMID: 24885074
- PMCID: PMC4234451
- DOI: 10.1021/jp503149x
Singlet oxygen generation on porous superhydrophobic surfaces: effect of gas flow and sensitizer wetting on trapping efficiency
Abstract
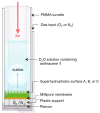
We describe physical-organic studies of singlet oxygen generation and transport into an aqueous solution supported on superhydrophobic surfaces on which silicon-phthalocyanine (Pc) particles are immobilized. Singlet oxygen ((1)O2) was trapped by a water-soluble anthracene compound and monitored in situ using a UV-vis spectrometer. When oxygen flows through the porous superhydrophobic surface, singlet oxygen generated in the plastron (i.e., the gas layer beneath the liquid) is transported into the solution within gas bubbles, thereby increasing the liquid-gas surface area over which singlet oxygen can be trapped. Higher photooxidation rates were achieved in flowing oxygen, as compared to when the gas in the plastron was static. Superhydrophobic surfaces were also synthesized so that the Pc particles were located in contact with, or isolated from, the aqueous solution to evaluate the relative effectiveness of singlet oxygen generated in solution and the gas phase, respectively; singlet oxygen generated on particles wetted by the solution was trapped more efficiently than singlet oxygen generated in the plastron, even in the presence of flowing oxygen gas. A mechanism is proposed that explains how Pc particle wetting, plastron gas composition and flow rate as well as gas saturation of the aqueous solution affect singlet oxygen trapping efficiency. These stable superhydrophobic surfaces, which can physically isolate the photosensitizer particles from the solution may be of practical importance for delivering singlet oxygen for water purification and medical devices.
Figures
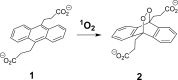




Similar articles
-
Superhydrophobic photosensitizers. Mechanistic studies of (1)O2 generation in the plastron and solid/liquid droplet interface.J Am Chem Soc. 2013 Dec 18;135(50):18990-8. doi: 10.1021/ja410529q. Epub 2013 Dec 10. J Am Chem Soc. 2013. PMID: 24295210 Free PMC article.
-
Superhydrophobic Photosensitizers: Airborne 1O2 Killing of an in Vitro Oral Biofilm at the Plastron Interface.ACS Appl Mater Interfaces. 2018 Aug 1;10(30):25819-25829. doi: 10.1021/acsami.8b09439. Epub 2018 Jul 18. ACS Appl Mater Interfaces. 2018. PMID: 29972022 Free PMC article.
-
Generating singlet oxygen bubbles: a new mechanism for gas-liquid oxidations in water.Langmuir. 2012 Feb 7;28(5):3053-60. doi: 10.1021/la204583v. Epub 2012 Jan 20. Langmuir. 2012. PMID: 22260325 Free PMC article.
-
Mimicking natural superhydrophobic surfaces and grasping the wetting process: a review on recent progress in preparing superhydrophobic surfaces.Adv Colloid Interface Sci. 2011 Dec 12;169(2):80-105. doi: 10.1016/j.cis.2011.08.005. Epub 2011 Sep 14. Adv Colloid Interface Sci. 2011. PMID: 21974918 Review.
-
Continuous Photo-Oxidation in a Vortex Reactor: Efficient Operations Using Air Drawn from the Laboratory.Org Process Res Dev. 2017 Jul 21;21(7):1042-1050. doi: 10.1021/acs.oprd.7b00153. Epub 2017 Jul 3. Org Process Res Dev. 2017. PMID: 28781513 Free PMC article. Review.
Cited by
-
Superhydrophobic Tipped Antimicrobial Photodynamic Therapy Device for the In Vivo Treatment of Periodontitis Using a Wistar Rat Model.ACS Appl Mater Interfaces. 2023 Nov 1;15(43):50083-50094. doi: 10.1021/acsami.3c12820. Epub 2023 Oct 20. ACS Appl Mater Interfaces. 2023. PMID: 37862708 Free PMC article.
-
A simple, inexpensive method for gas-phase singlet oxygen generation from sensitizer-impregnated filters: Potential application to bacteria/virus inactivation and pollutant degradation.Sci Total Environ. 2020 Dec 1;746:141186. doi: 10.1016/j.scitotenv.2020.141186. Epub 2020 Jul 23. Sci Total Environ. 2020. PMID: 32745862 Free PMC article.
-
Effect of treatment frequency on the efficacy of superhydrophobic antimicrobial photodynamic therapy of periodontitis in a wistar rat model.Photochem Photobiol. 2025 May-Jun;101(3):592-608. doi: 10.1111/php.14021. Epub 2024 Oct 10. Photochem Photobiol. 2025. PMID: 39387243
-
Evaluation of photosensitizer-containing superhydrophobic surfaces for the antibacterial treatment of periodontal biofilms.J Photochem Photobiol B. 2022 Aug;233:112458. doi: 10.1016/j.jphotobiol.2022.112458. Epub 2022 Apr 29. J Photochem Photobiol B. 2022. PMID: 35691161 Free PMC article.
-
High Singlet Oxygen Yields from a Polymer-Supported Photosensitizer via Superhydrophobicity and Control of Photosensitizer Morphology.Appl Surf Sci. 2025 Jun 30;695:162826. doi: 10.1016/j.apsusc.2025.162826. Epub 2025 Feb 28. Appl Surf Sci. 2025. PMID: 40538498
References
-
- Clennan E. L.; Pace A. Advances in Singlet Oxygen Chemistry. Tetrahedron 2005, 61, 6665–6691.
-
- Jirasek V.; Censky M.; Spalek O.; Kodymová J. High-Pressure Generator of Singlet Oxygen. Chem. Eng. Technol. 2013, 36, 1755–1763.
-
- Wootton R. C. R.; Fortt R.; deMello A. J. A Microfabricated Nanoreactor for Safe, Continuous Generation and Use of Singlet Oxygen. Org. Process Res. Dev. 2002, 6, 187–189.
-
- Coyle E. E.; Oelgemöller M. Micro-Photochemistry: Photochemistry in Microstructured Reactors. The New Photochemistry of the Future?. Photochem. Photobiol. Sci. 2008, 7, 1313–1322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

