Intermittent hypoxia-induced protein phosphatase 2A activation reduces PC12 cell proliferation and differentiation
- PMID: 24885237
- PMCID: PMC4058715
- DOI: 10.1186/1423-0127-21-46
Intermittent hypoxia-induced protein phosphatase 2A activation reduces PC12 cell proliferation and differentiation
Abstract
Background: Intermittent hypoxia (IH) plays a critical role in sleep breathing disorder-associated hippocampus impairments, including neurocognitive deficits, irreversible memory and learning impairments. IH-induced neuronal injury in the hippocampus may result from reduced precursor cell proliferation and the relative numbers of postmitotic differentiated neurons. However, the mechanisms underlying IH-induced reactive oxygen species (ROS) generation effects on cell proliferation and neuronal differentiation remain largely unknown.
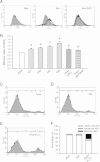
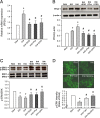
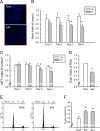
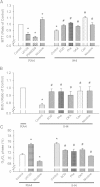
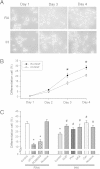
Results: ROS generation significantly increased after 1-4 days of IH without increased pheochromocytoma-12 (PC12) cell death, which resulted in increased protein phosphatase 2A (PP2A) mRNA and protein levels. After 3-4 days of IH, extracellular signal-regulated kinases 1/2 (ERK1/2) protein phosphorylation decreased, which could be reversed by superoxide dismutase (SOD), 1,10-phenanthroline (Phe), the PP2A phosphorylation inhibitors, okadaic acid (OKA) and cantharidin, and the ERK phosphorylation activator nicotine (p < 0.05). In particular, the significantly reduced cell proliferation and increased proportions of cells in the G0/G1 phase after 1-4 days of IH (p < 0.05), which resulted in decreased numbers of PC12 cells, could be reversed by treatment with SOD, Phe, PP2A inhibitors and an ERK activator. In addition, the numbers of nerve growth factor (NGF)-induced PC12 cells with neurite outgrowths after 3-4 days of IH were less than those after 4 days of RA, which was also reversed by SOD, Phe, PP2A inhibitors and an ERK activator.
Conclusions: Our results suggest that IH-induced ROS generation increases PP2A activation and subsequently downregulates ERK1/2 activation, which results in inhibition of PC12 cell proliferation through G0/G1 phase arrest and NGF-induced neuronal differentiation.
Figures
Similar articles
-
Reactive oxygen species, Ki-Ras, and mitochondrial superoxide dismutase cooperate in nerve growth factor-induced differentiation of PC12 cells.J Biol Chem. 2010 Jul 30;285(31):24141-53. doi: 10.1074/jbc.M109.098525. Epub 2010 May 21. J Biol Chem. 2010. PMID: 20495008 Free PMC article.
-
Hydrogen peroxide-induced neuronal apoptosis is associated with inhibition of protein phosphatase 2A and 5, leading to activation of MAPK pathway.Int J Biochem Cell Biol. 2009 Jun;41(6):1284-95. doi: 10.1016/j.biocel.2008.10.029. Epub 2008 Nov 6. Int J Biochem Cell Biol. 2009. PMID: 19038359
-
Pattern-specific sustained activation of tyrosine hydroxylase by intermittent hypoxia: role of reactive oxygen species-dependent downregulation of protein phosphatase 2A and upregulation of protein kinases.Antioxid Redox Signal. 2009 Aug;11(8):1777-89. doi: 10.1089/ars.2008.2368. Antioxid Redox Signal. 2009. PMID: 19335094 Free PMC article.
-
Dimethyloxalylglycine (DMOG), a Hypoxia Mimetic Agent, Does Not Replicate a Rat Pheochromocytoma (PC12) Cell Biological Response to Reduced Oxygen Culture.Biomolecules. 2022 Apr 3;12(4):541. doi: 10.3390/biom12040541. Biomolecules. 2022. PMID: 35454130 Free PMC article.
-
The protein phosphatase 2A regulatory subunits B'beta and B'delta mediate sustained TrkA neurotrophin receptor autophosphorylation and neuronal differentiation.Mol Cell Biol. 2009 Feb;29(3):662-74. doi: 10.1128/MCB.01242-08. Epub 2008 Nov 24. Mol Cell Biol. 2009. PMID: 19029245 Free PMC article.
Cited by
-
The role of lncRNA LSAT1 in the invasion and metastasis of non-small cell lung cancer under hypoxia.Transl Cancer Res. 2020 Feb;9(2):1125-1132. doi: 10.21037/tcr.2019.12.101. Transl Cancer Res. 2020. PMID: 35117456 Free PMC article.
-
Altered Wnt Signaling Pathway in Cognitive Impairment Caused by Chronic Intermittent Hypoxia: Focus on Glycogen Synthase Kinase-3β and β-catenin.Chin Med J (Engl). 2016 Apr 5;129(7):838-45. doi: 10.4103/0366-6999.178969. Chin Med J (Engl). 2016. PMID: 26996481 Free PMC article.
-
A Novel Chip for Cyclic Stretch and Intermittent Hypoxia Cell Exposures Mimicking Obstructive Sleep Apnea.Front Physiol. 2016 Jul 29;7:319. doi: 10.3389/fphys.2016.00319. eCollection 2016. Front Physiol. 2016. PMID: 27524971 Free PMC article.
-
Effect of different levels of intermittent hypoxia on autophagy of hippocampal neurons.Sleep Breath. 2017 Sep;21(3):791-798. doi: 10.1007/s11325-017-1512-7. Epub 2017 May 29. Sleep Breath. 2017. PMID: 28553681
-
Vagus nerve stimulation ameliorates cognitive impairment caused by hypoxia.Front Behav Neurosci. 2025 Jun 6;19:1555229. doi: 10.3389/fnbeh.2025.1555229. eCollection 2025. Front Behav Neurosci. 2025. PMID: 40546247 Free PMC article.
References
-
- Gozal E, Sachleben LR Jr, Rane MJ, Vega C, Gozal D. Mild sustained and intermittent hypoxia induce apoptosis in PC-12 cells via different mechanisms. Am J Physiol Cell Physiol. 2005;288(3):C535–C542. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

