Matrix metalloproteinase 12 is induced by heterogeneous nuclear ribonucleoprotein K and promotes migration and invasion in nasopharyngeal carcinoma
- PMID: 24885469
- PMCID: PMC4033617
- DOI: 10.1186/1471-2407-14-348
Matrix metalloproteinase 12 is induced by heterogeneous nuclear ribonucleoprotein K and promotes migration and invasion in nasopharyngeal carcinoma
Abstract
Background: Overexpression of heterogeneous nuclear ribonucleoprotein K (hnRNP K), a DNA/RNA binding protein, is associated with metastasis in nasopharyngeal carcinoma (NPC). However, the mechanisms underlying hnRNP K-mediated metastasis is unclear. The aim of the present study was to determine the role of matrix metalloproteinase (MMP) in hnRNP K-mediated metastasis in NPC.
Methods: We studied hnRNP K-regulated MMPs by analyzing the expression profiles of MMP family genes in NPC tissues and hnRNP K-knockdown NPC cells using Affymetrix microarray analysis and quantitative RT-PCR. The association of hnRNP K and MMP12 expression in 82 clinically proven NPC cases was determined by immunohistochemical analysis. The hnRNP K-mediated MMP12 regulation was determined by zymography and Western blot, as well as by promoter, DNA pull-down and chromatin immunoprecipitation (ChIP) assays. The functional role of MMP12 in cell migration and invasion was demonstrated by MMP12-knockdown and the treatment of MMP12-specific inhibitor, PF-356231.
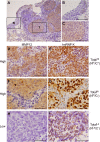
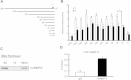
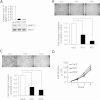
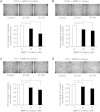
Results: MMP12 was overexpressed in NPC tissues, and this high level of expression was significantly correlated with high-level expression of hnRNP K (P = 0.026). The levels of mRNA, protein and enzyme activity of MMP12 were reduced in hnRNP K-knockdown NPC cells. HnRNP K interacting with the region spanning -42 to -33 bp of the transcription start site triggered transcriptional activation of the MMP12 promoter. Furthermore, inhibiting MMP12 by MMP12 knockdown and MMP12-specific inhibitor, PF-356231, significantly reduced the migration and invasion of NPC cells.
Conclusions: Overexpression of MMP12 was significantly correlated with hnRNP K in NPC tissues. HnRNP K can induce MMP12 expression and enzyme activity through activating MMP12 promoter, which promotes cell migration and invasion in NPC cells. In vitro experiments suggest that NPC metastasis with high MMP12 expression may be treated with PF-356231. HnRNP K and MMP12 may be potential therapeutic markers for NPC, but additional validation studies are warranted.
Figures


Similar articles
-
The antiapoptotic protein, FLIP, is regulated by heterogeneous nuclear ribonucleoprotein K and correlates with poor overall survival of nasopharyngeal carcinoma patients.Cell Death Differ. 2010 Sep;17(9):1463-73. doi: 10.1038/cdd.2010.24. Epub 2010 Mar 12. Cell Death Differ. 2010. PMID: 20224598
-
Function of AXL and molecular mechanisms in regulation of nasopharyngeal carcinoma.Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2022 Jun 28;47(6):685-697. doi: 10.11817/j.issn.1672-7347.2022.210786. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2022. PMID: 35837768 Free PMC article. Chinese, English.
-
Capn4 is a marker of poor clinical outcomes and promotes nasopharyngeal carcinoma metastasis via nuclear factor-κB-induced matrix metalloproteinase 2 expression.Cancer Sci. 2014 Jun;105(6):630-8. doi: 10.1111/cas.12416. Epub 2014 May 21. Cancer Sci. 2014. Retraction in: Cancer Sci. 2024 Oct;115(10):3491. doi: 10.1111/cas.16305. PMID: 24703594 Free PMC article. Retracted.
-
The Emerging Role of MMP12 in the Oral Environment.Int J Mol Sci. 2023 Feb 28;24(5):4648. doi: 10.3390/ijms24054648. Int J Mol Sci. 2023. PMID: 36902078 Free PMC article. Review.
-
Heterogeneous nuclear ribonucleoprotein G, nitric oxide, and oral carcinogenesis.Nitric Oxide. 2008 Sep;19(2):125-32. doi: 10.1016/j.niox.2008.04.012. Epub 2008 Apr 24. Nitric Oxide. 2008. PMID: 18474262 Review.
Cited by
-
Heterogeneous nuclear ribonucleoprotein K is associated with poor prognosis and regulates proliferation and apoptosis in bladder cancer.J Cell Mol Med. 2017 Jul;21(7):1266-1279. doi: 10.1111/jcmm.12999. Epub 2016 Nov 10. J Cell Mol Med. 2017. PMID: 27862976 Free PMC article.
-
ACVR1C/SMAD2 signaling promotes invasion and growth in retinoblastoma.Oncogene. 2019 Mar;38(12):2056-2075. doi: 10.1038/s41388-018-0543-2. Epub 2018 Nov 6. Oncogene. 2019. PMID: 30401983 Free PMC article.
-
Matrix metalloproteinase 13-containing exosomes promote nasopharyngeal carcinoma metastasis.Cancer Sci. 2015 Dec;106(12):1669-77. doi: 10.1111/cas.12818. Epub 2015 Oct 13. Cancer Sci. 2015. PMID: 26362844 Free PMC article.
-
The Pros and Cons of Estrogens in Prostate Cancer: An Update with a Focus on Phytoestrogens.Biomedicines. 2024 Jul 23;12(8):1636. doi: 10.3390/biomedicines12081636. Biomedicines. 2024. PMID: 39200101 Free PMC article. Review.
-
Serum MMP7, MMP10 and MMP12 level as negative prognostic markers in colon cancer patients.BMC Cancer. 2016 Jul 18;16:494. doi: 10.1186/s12885-016-2515-7. BMC Cancer. 2016. PMID: 27431388 Free PMC article.
References
-
- Barboro P, Repaci E, Rubagotti A, Salvi S, Boccardo S, Spina B, Truini M, Introini C, Puppo P, Ferrari N, Carmignani G, Boccardo F, Balbi C. Heterogeneous nuclear ribonucleoprotein K: altered pattern of expression associated with diagnosis and prognosis of prostate cancer. Br J Cancer. 2009;100(10):1608–1616. doi: 10.1038/sj.bjc.6605057. - DOI - PMC - PubMed
-
- Matta A, Tripathi SC, DeSouza LV, Grigull J, Kaur J, Chauhan SS, Srivastava A, Thakar A, Shukla NK, Duggal R, DattaGupta S, Ralhan R, MichaelSiu KW. Heterogeneous ribonucleoprotein K is a marker of oral leukoplakia and correlates with poor prognosis of squamous cell carcinoma. Int J Cancer. 2009;125(6):1398–1406. doi: 10.1002/ijc.24517. - DOI - PubMed
-
- Chen LC, Hsueh C, Tsang NM, Liang Y, Chang KP, Hao SP, Yu JS, Chang YS. Heterogeneous ribonucleoprotein k and thymidine phosphorylase are independent prognostic and therapeutic markers for nasopharyngeal carcinoma. Clin Cancer Res. 2008;14(12):3807–3813. doi: 10.1158/1078-0432.CCR-08-0155. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

