Viral persistence in colorectal cancer cells infected by Newcastle disease virus
- PMID: 24886301
- PMCID: PMC4030049
- DOI: 10.1186/1743-422X-11-91
Viral persistence in colorectal cancer cells infected by Newcastle disease virus
Abstract
Background: Newcastle disease virus (NDV), a single-stranded RNA virus of the family Paramyxoviridae, is a candidate virotherapy agent in cancer treatment. Promising responses were observed in clinical studies. Despite its high potential, the possibility of the virus to develop a persistent form of infection in cancer cells has not been investigated. Occurrence of persistent infection by NDV in cancer cells may cause the cells to be less susceptible to the virus killing. This would give rise to a population of cancer cells that remains viable and resistant to treatment.
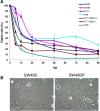
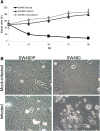
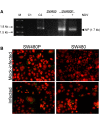
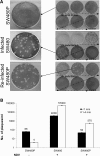
Results: During infection experiment in a series of colorectal cancer cell lines, we adventitiously observed a development of persistent infection by NDV in SW480 cells, but not in other cell lines tested. This cell population, designated as SW480P, showed resistancy towards NDV killing in a re-infection experiment. The SW480P cells retained NDV genome and produced virus progeny with reduced plaque forming ability.
Conclusion: These observations showed that NDV could develop persistent infection in cancer cells and this factor needs to be taken into consideration when using NDV in clinical settings.
Figures
Similar articles
-
Persistent Newcastle disease virus infection in bladder cancer cells is associated with putative pro-survival and anti-viral transcriptomic changes.BMC Cancer. 2021 May 27;21(1):625. doi: 10.1186/s12885-021-08345-y. BMC Cancer. 2021. PMID: 34044804 Free PMC article.
-
Genetic Modification of Oncolytic Newcastle Disease Virus for Cancer Therapy.J Virol. 2016 May 12;90(11):5343-5352. doi: 10.1128/JVI.00136-16. Print 2016 Jun 1. J Virol. 2016. PMID: 27009956 Free PMC article.
-
Newcastle Disease Virus Establishes Persistent Infection in Tumor Cells In Vitro: Contribution of the Cleavage Site of Fusion Protein and Second Sialic Acid Binding Site of Hemagglutinin-Neuraminidase.J Virol. 2017 Jul 27;91(16):e00770-17. doi: 10.1128/JVI.00770-17. Print 2017 Aug 15. J Virol. 2017. PMID: 28592535 Free PMC article.
-
Newcastle disease virus strain AF2240 as an oncolytic virus: A review.Acta Trop. 2018 Jul;183:126-133. doi: 10.1016/j.actatropica.2018.04.007. Epub 2018 Apr 4. Acta Trop. 2018. PMID: 29626432 Review.
-
Oncolytic Newcastle disease virus as a prospective anti-cancer therapy. A biologic agent with potential to break therapy resistance.Expert Opin Biol Ther. 2015;15(12):1757-71. doi: 10.1517/14712598.2015.1088000. Epub 2015 Oct 5. Expert Opin Biol Ther. 2015. PMID: 26436571 Review.
Cited by
-
Translocating shRNA: a novel approach to RNA interference with Newcastle disease virus as viral vector.J Gen Virol. 2025 Jul;106(7):002127. doi: 10.1099/jgv.0.002127. J Gen Virol. 2025. PMID: 40643574 Free PMC article.
-
Analysis of PPI networks of transcriptomic expression identifies hub genes associated with Newcastle disease virus persistent infection in bladder cancer.Sci Rep. 2023 May 5;13(1):7323. doi: 10.1038/s41598-022-20521-z. Sci Rep. 2023. PMID: 37147328 Free PMC article.
-
Metabolic Reprogramming in Colon Cancer Cells Persistently Infected with Newcastle Disease Virus.Cancers (Basel). 2023 Jan 28;15(3):811. doi: 10.3390/cancers15030811. Cancers (Basel). 2023. PMID: 36765769 Free PMC article.
-
Gene Expression Analysis of Human Papillomavirus-Associated Colorectal Carcinoma.Biomed Res Int. 2020 Mar 17;2020:5201587. doi: 10.1155/2020/5201587. eCollection 2020. Biomed Res Int. 2020. PMID: 32258125 Free PMC article. Clinical Trial.
-
Persistent Newcastle disease virus infection in bladder cancer cells is associated with putative pro-survival and anti-viral transcriptomic changes.BMC Cancer. 2021 May 27;21(1):625. doi: 10.1186/s12885-021-08345-y. BMC Cancer. 2021. PMID: 34044804 Free PMC article.
References
-
- Freeman AI, Zakay-Rones Z, Gomori JM, Linetsky E, Rasooly L, Greenbaum E, Rozenman-Yair S, Panet A, Libson E, Irving CS, Galun E, Siegal T. Phase I/II trial of intravenous NDV-HUJ oncolytic virus in recurrent glioblastoma multiforme. Mol Ther. 2006;13:221–228. doi: 10.1016/j.ymthe.2005.08.016. - DOI - PubMed
-
- Russell SJ, Peng KW, Bell JC. Oncolytic virotherapy. Nat Biotechnol. 2012;30:1–13.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

