Identification of mesenchymal stem cells and osteogenic factors in bone marrow aspirate and peripheral blood for spinal fusion by flow cytometry and proteomic analysis
- PMID: 24886437
- PMCID: PMC4037761
- DOI: 10.1186/1749-799X-9-32
Identification of mesenchymal stem cells and osteogenic factors in bone marrow aspirate and peripheral blood for spinal fusion by flow cytometry and proteomic analysis
Abstract
Background: An in vivo animal study and a prospective clinical study have indicated that bone marrow aspirate (BMA) augments spinal arthrodesis. However, there is no quantified data to explain why fusion rate can be augmented by BMA in lumbar posterolateral fusion.
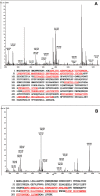
Methods: To analyze the proportion of mesenchymal stem cells (MSCs) and osteogenic factors in human BMA and peripheral blood (PB) of the same patient. Autologous BMA and PB from the patients were analyzed by flow cytometry (FACS) using cell markers for MSCs. The osteogenic potential of MSCs was determined by alkaline phosphatase (ALP) activity and calcium level quantification. Proteomics were used for the qualitative and quantitative mapping of the whole proteome from BMA and PB plasma. The mass-to-charge ratio was calculated by time-of-flight mass spectrometry (TOF-MS). The overexpression of protein was confirmed using Western blot analysis.
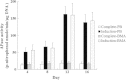
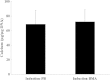
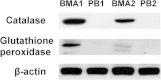
Results: The proportion of MSCs (CD34-/CD29+/CD105+) was higher in the BMA than that in the PB. Colony-forming cell (CFC) assays suggested that fewer colonies were formed in PB cultures than in BMA culture. There was no significant difference in the osteogenic potential of the MSCs between the PB and BMA. Proteomic mass spectrometry assays suggested that the levels of catalase (osteoclast inhibitor) and glutathione peroxidase 3 (osteogenic biomarker) were higher in the BMA than those in the PB, and this was confirmed by Western blot analysis.
Conclusions: The proportions of MSCs and osteogenic factors were higher in the BMA than in the PB. This may explain why fusion rate can be augmented by BMA in lumbar posterolateral fusion.
Figures



References
-
- Nather A. Use of allograft in spinal surgery. Ann Transpl. 1999;4:19–22. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

