Targeting the anionic region of human protease-activated receptor 4 inhibits platelet aggregation and thrombosis without interfering with hemostasis
- PMID: 24888424
- PMCID: PMC4127092
- DOI: 10.1111/jth.12619
Targeting the anionic region of human protease-activated receptor 4 inhibits platelet aggregation and thrombosis without interfering with hemostasis
Abstract
Background: Human platelet activation and aggregation is a complex process. To date, many therapies have been developed targeting proteins that mediate this process to prevent unwanted activation. However, the current standard of care for acute coronary syndromes still has limitations, including bleeding risk.
Objective: To evaluate the protease-activated receptor 4 (PAR4) anionic cluster as a viable antiplatelet target by using a polyclonal antibody (CAN12).
Methods: We used western blotting, aggregation and secretion ex vivo to evaluate the ability of CAN12 to interact with PAR4 and inhibit platelet activation. The effects of CAN12 in vivo were evaluated with the Rose Bengal arterial thrombosis model and two models of hemostasis.
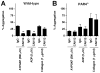
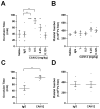
Results: CAN12 was able to interact with human PAR4 and delay PAR4 cleavage. In addition, CAN12 inhibited thrombin-induced human platelet aggregation and secretion in a dose-dependent manner. The specificity of CAN12 was agonist-dependent. In vivo, we determined that CAN12 was able to inhibit arterial thrombosis, and, using two independent methods, we found that CAN12 did not influence hemostasis.
Conclusion: Targeting the extracellular anionic cluster on PAR4 is a viable novel strategy as an antiplatelet therapy.
Keywords: G-protein-coupled receptors; antibodies; hemostasis; platelet aggregation inhibitors; protease-activated receptor-4 protein, human; thrombosis.
© 2014 International Society on Thrombosis and Haemostasis.
Conflict of interest statement
Disclosure of Conflict of Interest
The authors state that they have no conflicts of interest.
Figures

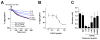



References
-
- Coughlin SR. Thrombin signalling and protease-activated receptors. Nature. 2000;407:258–64. - PubMed
-
- Steinhoff M, Buddenkotte J, Shpacovitch V, Rattenholl A, Moormann C, Vergnolle N, Luger TA, Hollenberg MD. Proteinase-activated receptors: transducers of proteinase-mediated signaling in inflammation and immune response. Endocr Rev. 2005;26:1–43. - PubMed
-
- Vu TK, Hung DT, Wheaton VI, Coughlin SR. Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanism of receptor activation. Cell. 1991;64:1057–68. - PubMed
-
- Nanevicz T, Ishii M, Wang L, Chen M, Chen J, Turck CW, Cohen FE, Coughlin SR. Mechanisms of thrombin receptor agonist specificity. Chimeric receptors and complementary mutations identify an agonist recognition site. J Biol Chem. 1995;270:21619–25. - PubMed
-
- Keularts IM, van Gorp RM, Feijge MA, Vuist WM, Heemskerk JW. alpha(2A)-adrenergic receptor stimulation potentiates calcium release in platelets by modulating cAMP levels. J Biol Chem. 2000;275:1763–72. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

