Myocyte-mediated arginase expression controls hyperargininemia but not hyperammonemia in arginase-deficient mice
- PMID: 24888478
- PMCID: PMC4428413
- DOI: 10.1038/mt.2014.99
Myocyte-mediated arginase expression controls hyperargininemia but not hyperammonemia in arginase-deficient mice
Abstract
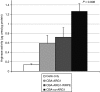
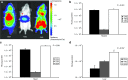
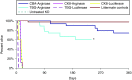
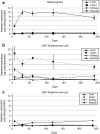
Human arginase deficiency is characterized by hyperargininemia and infrequent episodes of hyperammonemia that cause neurological impairment and growth retardation. We previously developed a neonatal mouse adeno-associated viral vector (AAV) rh10-mediated therapeutic approach with arginase expressed by a chicken β-actin promoter that controlled plasma ammonia and arginine, but hepatic arginase declined rapidly. This study tested a codon-optimized arginase cDNA and compared the chicken β-actin promoter to liver- and muscle-specific promoters. ARG1(-/-) mice treated with AAVrh10 carrying the liver-specific promoter also exhibited long-term survival and declining hepatic arginase accompanied by the loss of AAV episomes during subsequent liver growth. Although arginase expression in striated muscle was not expected to counteract hyperammonemia, due to muscle's lack of other urea cycle enzymes, we hypothesized that the postmitotic phenotype in muscle would allow vector genomes to persist, and hence contribute to decreased plasma arginine. As anticipated, ARG1(-/-) neonatal mice treated with AAVrh10 carrying a modified creatine kinase-based muscle-specific promoter did not survive longer than controls; however, their plasma arginine levels remained normal when animals were hyperammonemic. These data imply that plasma arginine can be controlled in arginase deficiency by muscle-specific expression, thus suggesting an alternative approach to utilizing the liver for treating hyperargininemia.
Figures

Similar articles
-
Minimal ureagenesis is necessary for survival in the murine model of hyperargininemia treated by AAV-based gene therapy.Gene Ther. 2015 Feb;22(2):111-5. doi: 10.1038/gt.2014.106. Epub 2014 Dec 4. Gene Ther. 2015. PMID: 25474440 Free PMC article.
-
Lethal phenotype in conditional late-onset arginase 1 deficiency in the mouse.Mol Genet Metab. 2013 Nov;110(3):222-30. doi: 10.1016/j.ymgme.2013.06.020. Epub 2013 Jul 6. Mol Genet Metab. 2013. PMID: 23920045 Free PMC article.
-
Long-term survival of the juvenile lethal arginase-deficient mouse with AAV gene therapy.Mol Ther. 2012 Oct;20(10):1844-51. doi: 10.1038/mt.2012.129. Epub 2012 Jul 3. Mol Ther. 2012. PMID: 22760543 Free PMC article.
-
Arginase-1 deficiency.J Mol Med (Berl). 2015 Dec;93(12):1287-96. doi: 10.1007/s00109-015-1354-3. Epub 2015 Oct 14. J Mol Med (Berl). 2015. PMID: 26467175 Review.
-
Hyperargininemia due to arginase I deficiency: the original patients and their natural history, and a review of the literature.Amino Acids. 2015 Sep;47(9):1751-62. doi: 10.1007/s00726-015-2032-z. Epub 2015 Jun 27. Amino Acids. 2015. PMID: 26123990 Review.
Cited by
-
Intermittent lipid nanoparticle mRNA administration prevents cortical dysmyelination associated with arginase deficiency.Mol Ther Nucleic Acids. 2022 Apr 27;28:859-874. doi: 10.1016/j.omtn.2022.04.012. eCollection 2022 Jun 14. Mol Ther Nucleic Acids. 2022. PMID: 35694211 Free PMC article.
-
Restoring Ureagenesis in Hepatocytes by CRISPR/Cas9-mediated Genomic Addition to Arginase-deficient Induced Pluripotent Stem Cells.Mol Ther Nucleic Acids. 2016 Nov 29;5(11):e394. doi: 10.1038/mtna.2016.98. Mol Ther Nucleic Acids. 2016. PMID: 27898091 Free PMC article.
-
Rescue of the Functional Alterations of Motor Cortical Circuits in Arginase Deficiency by Neonatal Gene Therapy.J Neurosci. 2016 Jun 22;36(25):6680-90. doi: 10.1523/JNEUROSCI.0897-16.2016. J Neurosci. 2016. PMID: 27335400 Free PMC article.
-
Gene therapy for guanidinoacetate methyltransferase deficiency restores cerebral and myocardial creatine while resolving behavioral abnormalities.Mol Ther Methods Clin Dev. 2022 Mar 28;25:278-296. doi: 10.1016/j.omtm.2022.03.015. eCollection 2022 Jun 9. Mol Ther Methods Clin Dev. 2022. PMID: 35505663 Free PMC article.
-
Hepatic arginase deficiency fosters dysmyelination during postnatal CNS development.JCI Insight. 2019 Sep 5;4(17):e130260. doi: 10.1172/jci.insight.130260. JCI Insight. 2019. PMID: 31484827 Free PMC article.
References
-
- Jenkinson CP, Grody WW, Cederbaum SD. Comparative properties of arginases. Comp Biochem Physiol B, Biochem Mol Biol. 1996;114:107–132. - PubMed
-
- Mori M, Gotoh T. Regulation of nitric oxide production by arginine metabolic enzymes. Biochem Biophys Res Commun. 2000;275:715–719. - PubMed
-
- Mori M. Regulation of nitric oxide synthesis and apoptosis by arginase and arginine recycling. J Nutr. 2007;137 suppl. 2:1616S–1620S. - PubMed
-
- Kepka-Lenhart D, Mistry SK, Wu G, Morris SM., Jr Arginase I: a limiting factor for nitric oxide and polyamine synthesis by activated macrophages. Am J Physiol Regul Integr Comp Physiol. 2000;279:R2237–R2242. - PubMed
-
- Iyer R, Jenkinson CP, Vockley JG, Kern RM, Grody WW, Cederbaum S. The human arginases and arginase deficiency. J Inherit Metab Dis. 1998;21 suppl. 1:86–100. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

