Phosphorus centers of different hybridization in phosphaalkene-substituted phospholes
- PMID: 24890504
- PMCID: PMC4506554
- DOI: 10.1002/chem.201402406
Phosphorus centers of different hybridization in phosphaalkene-substituted phospholes
Abstract
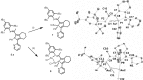
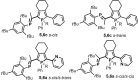
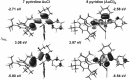
Phosphole-substituted phosphaalkenes (PPAs) of the general formula Mes*P=C(CH(3))-(C(4)H(2)P(Ph))-R 5 a-c (Mes*=2,4,6-tBu(3)Ph; R=2-pyridyl (a), 2-thienyl (b), phenyl (c)) have been prepared from octa-1,7-diyne-substituted phosphaalkenes by utilizing the Fagan-Nugent route. The presence of two differently hybridized phosphorus centers (σ(2) ,λ(3) and σ(3) ,λ(3)) in 5 offers the possibility to selectively tune the HOMO-LUMO gap of the compounds by utilizing the different reactivity of the two phosphorus heteroatoms. Oxidation of 5 a-c by sulfur proceeds exclusively at the σ(3) ,λ(3) -phosphorus atom, thus giving rise to the corresponding thioxophospholes 6 a-c. Similarly, 5 a is selectively coordinated by AuCl at the σ(3),λ(3) -phosphorus atom. Subsequent second AuCl coordination at the σ(2),λ(3) -phosphorus heteroatom results in a dimetallic species that is characterized by a gold-gold interaction that provokes a change in π conjugation. Spectroscopic, electrochemical, and theoretical investigations show that the phosphaalkene and the phosphole both have a sizable impact on the electronic properties of the compounds. The presence of the phosphaalkene unit induces a decrease of the HOMO-LUMO gap relative to reference phosphole-containing π systems that lack a P=C substituent.
Keywords: X-ray diffraction; conjugation; electronic structure; phosphaalkenes; phosphorus.
© 2014 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures





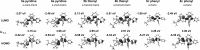
Similar articles
-
Synthesis, electronic properties, and reactivity of phospholes and 1,1'-biphospholes bearing 2- or 3-thienyl C-substituents.Chemistry. 2009;15(19):4914-24. doi: 10.1002/chem.200802677. Chemistry. 2009. PMID: 19301337
-
3,4-Dithiaphosphole and 3,3',4,4'-tetrathia-1,1'-biphosphole π-conjugated systems: S makes the impact.Chemistry. 2010 Oct 4;16(37):11340-56. doi: 10.1002/chem.201001463. Chemistry. 2010. PMID: 20715213
-
Acetylene-expanded dendralene segments with exotopic phosphaalkene units.Chemistry. 2011 Oct 17;17(43):12153-62. doi: 10.1002/chem.201101358. Epub 2011 Sep 14. Chemistry. 2011. PMID: 21915927
-
pi-Conjugated phosphole derivatives: synthesis, optoelectronic functions and coordination chemistry.Dalton Trans. 2008 Dec 28;(48):6865-76. doi: 10.1039/b810976a. Epub 2008 Nov 3. Dalton Trans. 2008. PMID: 19050770 Review.
-
Main Group Pentafulvenes: Challenges and Opportunities in Heavy Main Group Isolobal Substitution of Pentafulvene.Chemphyschem. 2024 Aug 1;25(15):e202300940. doi: 10.1002/cphc.202300940. Epub 2024 Jun 27. Chemphyschem. 2024. PMID: 38709950 Review.
Cited by
-
Cooperative Gold Nanoparticle Stabilization by Acetylenic Phosphaalkenes.Angew Chem Int Ed Engl. 2015 Sep 1;54(36):10634-8. doi: 10.1002/anie.201504834. Epub 2015 Jul 23. Angew Chem Int Ed Engl. 2015. PMID: 26211907 Free PMC article.
-
Golden Age of Fluorenylidene Phosphaalkenes-Synthesis, Structures, and Optical Properties of Heteroaromatic Derivatives and Their Gold Complexes.J Org Chem. 2020 Nov 20;85(22):14619-14626. doi: 10.1021/acs.joc.0c01336. Epub 2020 Sep 4. J Org Chem. 2020. PMID: 32830965 Free PMC article.
References
-
- Haley MM, Tykwinski RR. Carbon-Rich Compounds. Weinheim: Wiley-VCH; 2006.
-
- Diederich F, Stang PJ, Tykwinski RR. Acetylene Chemistry. Weinheim: Wiley-VCH; 2005.
-
- Tour JM. Molecular Electronics, Commercial Insights, Chemistry, Devices, Architecture and Programming. Singapore: World Scientific Publishing; 2003.
-
- Müllen K, Scherf U. Organic Light Emitting Devices: Synthesis Properties and Applications. Weinheim: Wiley-VCH; 2006.
-
- Usta H, Facchetti A, Marks TJ. Acc. Chem. Res. 2011;44:501–510. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

