Preclinical studies of amixicile, a systemic therapeutic developed for treatment of Clostridium difficile infections that also shows efficacy against Helicobacter pylori
- PMID: 24890599
- PMCID: PMC4136022
- DOI: 10.1128/AAC.03112-14
Preclinical studies of amixicile, a systemic therapeutic developed for treatment of Clostridium difficile infections that also shows efficacy against Helicobacter pylori
Abstract
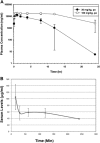
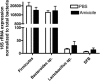
Amixicile shows efficacy in the treatment of Clostridium difficile infections (CDI) in a mouse model, with no recurrence of CDI. Since amixicile selectively inhibits the action of a B vitamin (thiamine pyrophosphate) cofactor of pyruvate:ferredoxin oxidoreductase (PFOR), it may both escape mutation-based drug resistance and spare beneficial probiotic gut bacteria that do not express this enzyme. Amixicile is a water-soluble derivative of nitazoxanide (NTZ), an antiparasitic therapeutic that also shows efficacy against CDI in humans. In comparative studies, amixicile showed no toxicity to hepatocytes at 200 μM (NTZ was toxic above 10 μM); was not metabolized by human, dog, or rat liver microsomes; showed equivalence or superiority to NTZ in cytochrome P450 assays; and did not activate efflux pumps (breast cancer resistance protein, P glycoprotein). A maximum dose (300 mg/kg) of amixicile given by the oral or intraperitoneal route was well tolerated by mice and rats. Plasma exposure (rats) based on the area under the plasma concentration-time curve was 79.3 h · μg/ml (30 mg/kg dose) to 328 h · μg/ml (100 mg/kg dose), the maximum concentration of the drug in serum was 20 μg/ml, the time to the maximum concentration of the drug in serum was 0.5 to 1 h, and the half-life was 5.6 h. Amixicile did not concentrate in mouse feces or adversely affect gut populations of Bacteroides species, Firmicutes, segmented filamentous bacteria, or Lactobacillus species. Systemic bioavailability was demonstrated through eradication of Helicobacter pylori in a mouse infection model. In summary, the efficacy of amixicile in treating CDI and other infections, together with low toxicity, an absence of mutation-based drug resistance, and excellent drug metabolism and pharmacokinetic metrics, suggests a potential for broad application in the treatment of infections caused by PFOR-expressing microbial pathogens in addition to CDI.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures

References
-
- Cohen SH, Gerding DN, Johnson S, Kelly CP, Loo VG, McDonald LC, Pepin J, Wilcox MH. 2010. Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Infect. Control Hosp. Epidemiol. 31:431–455. 10.1086/651706 - DOI - PubMed
-
- See I, Mu Y, Cohen J, Beldavs ZG, Winston LG, Dumyati G, Holzbauer S, Dunn J, Farley MM, Lyons C, Johnston H, Phipps E, Perlmutter R, Anderson L, Gerding DN, Lessa FC. 2014. NAP1 strain type predicts outcomes from Clostridium difficile infection. Clin. Infect. Dis. 58:1394–1400. 10.1093/cid/ciu125 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

