Tiotropium Respimat® in asthma: a double-blind, randomised, dose-ranging study in adult patients with moderate asthma
- PMID: 24890738
- PMCID: PMC4066691
- DOI: 10.1186/1465-9921-15-61
Tiotropium Respimat® in asthma: a double-blind, randomised, dose-ranging study in adult patients with moderate asthma
Abstract
Background: Tiotropium, a once-daily long-acting anticholinergic bronchodilator, when administered via Respimat® SoftMist™ inhaler (tiotropium Respimat®) significantly reduces the risk of severe exacerbations and improves lung function in patients with severe persistent asthma that is not fully controlled despite using inhaled corticosteroids (ICS) and long-acting β2-agonists. To further explore the dose-response curve in asthma, we investigated the efficacy and safety of three different doses of tiotropium Respimat® as add-on to ICS in symptomatic patients with moderate persistent asthma.
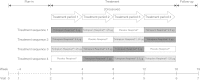
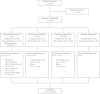
Methods: In this randomised, double-blind, placebo-controlled, four-way crossover study, patients were randomised to tiotropium Respimat® 5 μg, 2.5 μg or 1.25 μg or placebo Respimat®, once daily in the evening. Each treatment was administered for 4 weeks, without washout between treatment periods. Eligibility criteria included ≥60% and ≤90% of predicted normal forced expiratory volume in 1 second (FEV1) and seven-question Asthma Control Questionnaire mean score of ≥1.5. Patients were required to continue maintenance treatment with stable medium-dose ICS for at least 4 weeks prior to and during the treatment period. Long-acting β2-agonists were not permitted during the treatment phase. The primary efficacy end point was peak FEV1 measured within 3 hours after dosing (peak FEV1(0-3h)) at the end of each 4-week period, analysed as a response (change from study baseline).
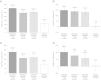
Results: In total, 149 patients were randomised and 141 completed the study. Statistically significant improvements in peak FEV1(0-3h) response were observed with each tiotropium Respimat® dose versus placebo (all P < 0.0001). The largest difference from placebo was with tiotropium Respimat® 5 μg (188 mL). Trough FEV1 and FEV1 area under the curve (AUC)(0-3h) responses were greater with each tiotropium Respimat® dose than with placebo (all P < 0.0001), and both were greatest with 5 μg. Peak forced vital capacity (FVC)(0-3h), trough FVC and FVC AUC(0-3h) responses, versus placebo, were greatest with tiotropium Respimat® 5 μg (P < 0.0001, P = 0.0012 and P < 0.0001, respectively). Incidence of adverse events was comparable between placebo and all tiotropium Respimat® groups.
Conclusions: Once-daily tiotropium Respimat® add-on to medium-dose ICS improves lung function in symptomatic patients with moderate asthma. Overall, improvements were largest with tiotropium Respimat® 5 μg.
Trial registration: ClinicalTrials.gov identifier NCT01233284.
Figures
References
-
- Global Initiative for Asthma. Global strategy for asthma management and prevention. Revised 2014. [ http://www.ginasthma.org/local/uploads/files/GINA_Report_2014.pdf]
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

