A Phase II study of bevacizumab in combination with trastuzumab and docetaxel in HER2 positive metastatic breast cancer
- PMID: 24894652
- PMCID: PMC4303337
- DOI: 10.1007/s10637-014-0122-5
A Phase II study of bevacizumab in combination with trastuzumab and docetaxel in HER2 positive metastatic breast cancer
Abstract
Background: Preclinical and early clinical data support the use of Vascular Epithelial Growth Factor (VEGF)-targeted therapy with trastuzumab in Human Epidermal Receptor 2 (HER2) positive breast cancer. Adding bevacizumab to a taxane (docetaxel or paclitaxel) improves progression free survival (PFS) of metastatic breast cancer (MBC) patients.
Objectives: We evaluated the efficacy and feasibility of combining bevacizumab with trastuzumab and docetaxel in patients with HER2- positive MBC who received 0-1 prior chemotherapy regimens for metastatic disease. The primary end point was PFS.
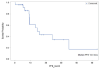
Materials and methods: Eligible patients received bevacizumab (15 mg/kg), trastuzumab (8 mg/kg loading dose followed by 6 mg/kg), and docetaxel (100 mg/m2 initially, later amended to 75 mg/m2) every three weeks for six cycles and then were allowed to receive bevacizumab and trastuzumab alone. Results Thirteen (50%) of 26 patients enrolled completed all 6 cycles of bevacizumab, trastuzumab and docetaxel and went on to receive bevacizumab and trastuzumab alone (median: 11 cycles). The most common grade 3 or 4 toxicities include: neutropenia (8%), septic death (4%), infection not associated with neutropenia (15%), fatigue (27%), mylagia and/or arthraligia (20%), and hand-foot syndrome (8%). One patient (4%) and six patients (23%) developed grade 3 and grade 2 hypertension, respectively. Two (8%) patients had transient grade 2 drop in Left Ventricular Ejection Fraction (LVEF) with full recovery later. The median progression free survival (PFS) was 14.3 months (95% CI: 9.3-35 months), the objective response rate (ORR), defined as the best response of complete response (CR) or partial response (PR) was (12/26) 46%. The clinical benefit rate (CBR), defined as the best response of CR or PR or stable disease (SD) for at least 24 weeks, was (18/26) 69% (95% CI: 48-86%).
Conclusion: The combination of bevacizumab, trastuzumab and docetaxel is well tolerated and is clinically active in patients with HER2-positive MBC, with response rate and PFS comparable to previous reports utilizing higher dose of docetaxel (100 mg/m2). Recent randomized trials did not demonstrate additional overall survival (OS) benefit of adding bevacizumab to trastuzumab and docetaxel despite an improvement in PFS. Identification of predictive biomarkers and careful patient selection should be incorporated in further investigation of anti-VEGF in breast cancer.
Figures
Similar articles
-
AVEREL: a randomized phase III Trial evaluating bevacizumab in combination with docetaxel and trastuzumab as first-line therapy for HER2-positive locally recurrent/metastatic breast cancer.J Clin Oncol. 2013 May 10;31(14):1719-25. doi: 10.1200/JCO.2012.44.7912. Epub 2013 Apr 8. J Clin Oncol. 2013. PMID: 23569311 Clinical Trial.
-
Phase II multicenter study of docetaxel and bevacizumab with or without trastuzumab as first-line treatment for patients with metastatic breast cancer.Clin Breast Cancer. 2014 Jun;14(3):161-8. doi: 10.1016/j.clbc.2013.12.003. Epub 2013 Dec 27. Clin Breast Cancer. 2014. PMID: 24566467 Clinical Trial.
-
Preliminary safety and efficacy of first-line pertuzumab combined with trastuzumab and taxane therapy for HER2-positive locally recurrent or metastatic breast cancer (PERUSE).Ann Oncol. 2019 May 1;30(5):766-773. doi: 10.1093/annonc/mdz061. Ann Oncol. 2019. PMID: 30796821 Clinical Trial.
-
Effect of pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer: A meta-analysis .Int J Clin Pharmacol Ther. 2017 Sep;55(9):720-727. doi: 10.5414/CP202921. Int J Clin Pharmacol Ther. 2017. PMID: 28737130
-
Bevacizumab in combination with a taxane for the first-line treatment of HER2-negative metastatic breast cancer.Health Technol Assess. 2011 May;15 Suppl 1:1-12. doi: 10.3310/hta15suppl1/01. Health Technol Assess. 2011. PMID: 21609648 Review.
Cited by
-
Recent advances in systemic therapy: Advances in systemic therapy for HER2-positive metastatic breast cancer.Breast Cancer Res. 2009;11(4):207. doi: 10.1186/bcr2324. Epub 2009 Jul 15. Breast Cancer Res. 2009. PMID: 19664181 Free PMC article. Review.
-
Selective targeting of liver cancer with the endothelial marker CD146.Oncotarget. 2014 Sep 30;5(18):8614-24. doi: 10.18632/oncotarget.2345. Oncotarget. 2014. PMID: 25238265 Free PMC article.
-
Pilot study of bevacizumab in combination with docetaxel and cyclophosphamide as adjuvant treatment for patients with early stage HER-2 negative breast cancer, including analysis of candidate circulating markers of cardiac toxicity: ICORG 08-10 trial.Ther Adv Med Oncol. 2019 Jul 24;11:1758835919864236. doi: 10.1177/1758835919864236. eCollection 2019. Ther Adv Med Oncol. 2019. PMID: 31384312 Free PMC article.
-
Current Therapies for Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer Patients.Front Oncol. 2018 Apr 3;8:89. doi: 10.3389/fonc.2018.00089. eCollection 2018. Front Oncol. 2018. PMID: 29670855 Free PMC article. Review.
-
Clinical effects of prior trastuzumab on combination eribulin mesylate plus trastuzumab as first-line treatment for human epidermal growth factor receptor 2 positive locally recurrent or metastatic breast cancer: results from a Phase II, single-arm, multicenter study.Breast Cancer (Dove Med Press). 2016 Dec 7;8:231-239. doi: 10.2147/BCTT.S98696. eCollection 2016. Breast Cancer (Dove Med Press). 2016. PMID: 27994483 Free PMC article.
References
-
- Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344(11):783–792. doi:10.1056/nejm200103153441101. - PubMed
-
- Marty M, Cognetti F, Maraninchi D, Snyder R, Mauriac L, Tubiana-Hulin M, Chan S, Grimes D, Anton A, Lluch A, Kennedy J, O’Byrne K, Conte P, Green M, Ward C, Mayne K, Extra JM. Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. J Clin Oncol Off J Am Soc Clin Oncol. 2005;23(19):4265–4274. doi:10.1200/jco.2005.04.173. - PubMed
-
- Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353(16):1673–1684. doi:10.1056/NEJMoa052122. - PubMed
-
- Dang C, Fornier M, Sugarman S, Troso-Sandoval T, Lake D, D’Andrea G, Seidman A, Sklarin N, Dickler M, Currie V, Gilewski T, Moynahan ME, Drullinsky P, Robson M, Wasserheit-Leiblich C, Mills N, Steingart R, Panageas K, Norton L, Hudis C. The safety of dose-dense doxorubicin and cyclophosphamide followed by paclitaxel with trastuzumab in HER-2/neu overexpressed/amplified breast cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2008;26(8):1216–1222. doi:10.1200/jco.2007.12.0733. - PubMed
-
- Yen L, You XL, Al Moustafa AE, Batist G, Hynes NE, Mader S, Meloche S, Alaoui-Jamali MA. Heregulin selectively upregulates vascular endothelial growth factor secretion in cancer cells and stimulates angiogenesis. Oncogene. 2000;19(31):3460–3469. doi:10.1038/sj.onc.1203685. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous