Activation and inhibition of transient receptor potential TRPM3-induced gene transcription
- PMID: 24895737
- PMCID: PMC4009006
- DOI: 10.1111/bph.12524
Activation and inhibition of transient receptor potential TRPM3-induced gene transcription
Abstract
Background and purpose: Transient receptor potential-3 (TRPM3) channels function as Ca2+ permeable cation channels. While the natural ligands for these channels are still unknown, several compounds have been described that either activate or inhibit TRPM3 channel activity. experimental approach: We assessed TRPM3-mediated gene transcription, which relies on the induction of intracellular signalling to the nucleus following activation of TRPM3 channels. Activator protein-1 (AP-1) and Egr-1-responsive reporter genes were integrated into the chromatin of the cells. This strategy enabled us to analyse gene transcription of the AP-1 and Egr-1-responsive reporter genes that were packed into an ordered chromatin structure.
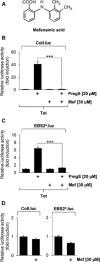
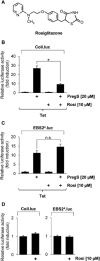
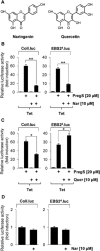
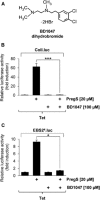
Key results: The neurosteroid pregnenolone sulfate strikingly up-regulated AP-1 and Egr-1 transcriptional activity, while nifedipine and D-erythro-sphingosine, also putative activators of TRPM3 channels, exhibited either no or TRPM3-independent effects on gene transcription. In addition, pregnenolone sulfate robustly enhanced the transcriptional activation potential of the ternary complex factor Elk-1. Pregnenolone sulfate-induced activation of gene transcription was blocked by treatment with mefenamic acid and, to a lesser extent, by the polyphenol naringenin. In contrast, progesterone, pregnenolone and rosiglitazone reduced AP-1 activity in the cells, but had no inhibitory effect on Egr-1 activity in pregnenolone sulfate-stimulated cells.
Conclusion and implications: Pregnenolone sulfate is a powerful activator of TRPM3-mediated gene transcription, while transcription is completely inhibited by mefenamic acid in cells expressing activated TRPM3 channels. Both compounds are valuable tools for further investigating the biological functions of TRPM3 channels.
Figures
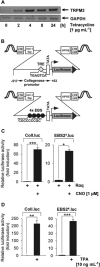
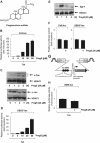
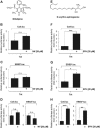
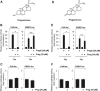

References
-
- Al-Sarraj A, Thiel G. Substance P induced biosynthesis of the zinc finger transcription factor Egr-1 in human glioma cells requires activation of the EGF receptor and of extracellular signal-regulated protein kinase. Neurosci Lett. 2002;332:111–114. - PubMed
-
- Al-Sarraj A, Thiel G. The zinc finger transcription factor Egr-1 is upregulated in arsenite-treated human keratinocytes. J Mol Med. 2004;82:530–538. - PubMed
-
- Bauer I, Al Sarraj J, Vinson C, Larsen R, Thiel G. Interleukin-1β and tetradecanoylphorbol acetate-induced biosynthesis of tumor necrosis factor α in human hepatoma cells involves the transcription factors ATF2 and c-Jun and stress-activated protein kinases. J Cell Biochem. 2007;100:242–255. - PubMed
-
- Bauer I, Hohl M, Al-Sarraj A, Vinson C, Thiel G. Transcriptional activation of the Egr-1 gene mediated by tetradecanoylphorbol acetate and extracellular signal-regulated protein kinase. Arch Biochem Biophys. 2005;438:36–52. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

