Control of the nigrostriatal dopamine neuron activity and motor function by the tail of the ventral tegmental area
- PMID: 24896615
- PMCID: PMC4200489
- DOI: 10.1038/npp.2014.129
Control of the nigrostriatal dopamine neuron activity and motor function by the tail of the ventral tegmental area
Abstract
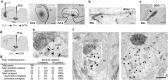
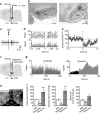
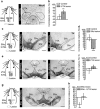
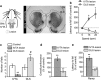
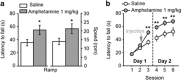
Midbrain dopamine neurons are implicated in various psychiatric and neurological disorders. The GABAergic tail of the ventral tegmental area (tVTA), also named the rostromedial tegmental nucleus (RMTg), displays dense projections to the midbrain and exerts electrophysiological control over dopamine cells of the VTA. However, the influence of the tVTA on the nigrostriatal pathway, from the substantia nigra pars compacta (SNc) to the dorsal striatum, and on related functions remains to be addressed. The present study highlights the role played by the tVTA as a GABA brake for the nigrostriatal system, demonstrating a critical influence over motor functions. Using neuroanatomical approaches with tract tracing and electron microscopy, we reveal the presence of a tVTA-SNc-dorsal striatum pathway. Using in vivo electrophysiology, we prove that the tVTA is a major inhibitory control center for SNc dopamine cells. Using behavioral approaches, we demonstrate that the tVTA controls rotation behavior, motor coordination, and motor skill learning. The motor enhancements observed after ablation of the tVTA are in this regard comparable with the performance-enhancing properties of amphetamine, a drug used in doping. These findings demonstrate that the tVTA is a major GABA brake for nigral dopamine systems and nigrostriatal functions, and they raise important questions about how the tVTA is integrated within the basal ganglia circuitry. They also warrant further research on the tVTA's role in motor and dopamine-related pathological contexts such as Parkinson's disease.
Figures

References
-
- Albin RL, Young AB, Penney JB. The functional anatomy of basal ganglia disorders. Trends Neurosci. 1989;12:366–375. - PubMed
-
- Akita H, Ogata M, Jitsuki S, Ogura T, Oh-Nishi A, Hoka S, et al. Nigral injection of antisense oligonucleotides to synaptotagmin I using HVJ-liposome vectors causes disruption of dopamine release in the striatum and impaired skill learning. Brain Res. 2006;1095:178–189. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

