Biotin analogues with antibacterial activity are potent inhibitors of biotin protein ligase
- PMID: 24900501
- PMCID: PMC4025796
- DOI: 10.1021/ml300106p
Biotin analogues with antibacterial activity are potent inhibitors of biotin protein ligase
Abstract
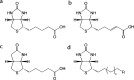
There is a desperate need to develop new antibiotic agents to combat the rise of drug-resistant bacteria, such as clinically important Staphylococcus aureus. The essential multifunctional enzyme, biotin protein ligase (BPL), is one potential drug target for new antibiotics. We report the synthesis and characterization of a series of biotin analogues with activity against BPLs from S. aureus, Escherichia coli, and Homo sapiens. Two potent inhibitors with K i < 100 nM were identified with antibacterial activity against a panel of clinical isolates of S. aureus (MIC 2-16 μg/mL). Compounds with high ligand efficiency and >20-fold selectivity between the isozymes were identified and characterized. The antibacterial mode of action was shown to be via inhibition of BPL. The bimolecular interactions between the BPL and the inhibitors were defined by surface plasmon resonance studies and X-ray crystallography. These findings pave the way for second-generation inhibitors and antibiotics with greater potency and selectivity.
Keywords: antibiotic; biotin protein ligase; enzyme; enzyme inhibitor; medicinal chemistry.
Figures


Similar articles
-
Biotin Protein Ligase Is a Target for New Antibacterials.Antibiotics (Basel). 2016 Jul 25;5(3):26. doi: 10.3390/antibiotics5030026. Antibiotics (Basel). 2016. PMID: 27463729 Free PMC article. Review.
-
Selective inhibition of biotin protein ligase from Staphylococcus aureus.J Biol Chem. 2012 May 18;287(21):17823-17832. doi: 10.1074/jbc.M112.356576. Epub 2012 Mar 21. J Biol Chem. 2012. PMID: 22437830 Free PMC article.
-
Advanced Resistance Studies Identify Two Discrete Mechanisms in Staphylococcus aureus to Overcome Antibacterial Compounds that Target Biotin Protein Ligase.Antibiotics (Basel). 2020 Apr 6;9(4):165. doi: 10.3390/antibiotics9040165. Antibiotics (Basel). 2020. PMID: 32268615 Free PMC article.
-
Improved Synthesis of Biotinol-5'-AMP: Implications for Antibacterial Discovery.ACS Med Chem Lett. 2014 Dec 11;6(2):216-20. doi: 10.1021/ml500475n. eCollection 2015 Feb 12. ACS Med Chem Lett. 2014. PMID: 25699152 Free PMC article.
-
Structure guided design of biotin protein ligase inhibitors for antibiotic discovery.Curr Top Med Chem. 2014;14(1):4-20. doi: 10.2174/1568026613666131111103149. Curr Top Med Chem. 2014. PMID: 24236729 Review.
Cited by
-
Bisubstrate Inhibitors of Biotin Protein Ligase in Mycobacterium tuberculosis Resistant to Cyclonucleoside Formation.ACS Med Chem Lett. 2013 Dec 12;4(12):1213-7. doi: 10.1021/ml400328a. ACS Med Chem Lett. 2013. PMID: 24363833 Free PMC article.
-
Fighting Antimicrobial Resistance: Innovative Drugs in Antibacterial Research.Angew Chem Int Ed Engl. 2025 Mar 3;64(10):e202414325. doi: 10.1002/anie.202414325. Epub 2025 Feb 10. Angew Chem Int Ed Engl. 2025. PMID: 39611429 Free PMC article. Review.
-
Multiple Ehrlichia chaffeensis Genes Critical for Its Persistent Infection in a Vertebrate Host Are Identified by Random Mutagenesis Coupled with In Vivo Infection Assessment.Infect Immun. 2020 Sep 18;88(10):e00316-20. doi: 10.1128/IAI.00316-20. Print 2020 Sep 18. Infect Immun. 2020. PMID: 32747600 Free PMC article.
-
Chloromethyl-triazole: a new motif for site-selective pseudo-acylation of proteins.Chem Commun (Camb). 2016 Oct 6;52(82):12230-12232. doi: 10.1039/c6cc06801d. Chem Commun (Camb). 2016. PMID: 27722332 Free PMC article.
-
Biotin Protein Ligase Is a Target for New Antibacterials.Antibiotics (Basel). 2016 Jul 25;5(3):26. doi: 10.3390/antibiotics5030026. Antibiotics (Basel). 2016. PMID: 27463729 Free PMC article. Review.
References
-
- Cooper M. A.; Shlaes D. Fix the antibiotics pipeline. Nature 2011, 472734132. - PubMed
-
- Allegranzi B.; Bagheri Nejad S.; Combescure C.; Graafmans W.; Attar H.; Donaldson L.; Pittet D. Burden of endemic health-care-associated infection in developing countries: Systematic review and meta-analysis. Lancet 2011, 3779761228–241. - PubMed
-
- Jean S. S.; Hsueh P. R. High burden of antimicrobial resistance in Asia. Int. J. Antimicrob. Agents 2011, 374291–295. - PubMed
-
- Polyak S. W.; Abell A. D.; Wilce M. C.; Zhang L.; Booker G. W. Structure, function and selective inhibition of bacterial acetyl-CoA carboxylase. Appl. Microbiol. Biotechnol. 2012, 933983–992. - PubMed
LinkOut - more resources
Full Text Sources
Chemical Information

