Retreatment with varenicline for smoking cessation in smokers who have previously taken varenicline: a randomized, placebo-controlled trial
- PMID: 24911368
- PMCID: PMC4151018
- DOI: 10.1038/clpt.2014.124
Retreatment with varenicline for smoking cessation in smokers who have previously taken varenicline: a randomized, placebo-controlled trial
Abstract
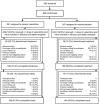
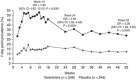
The efficacy and safety of retreatment with varenicline in smokers attempting to quit were evaluated in this randomized, double-blind, placebo-controlled, multicenter trial (Australia, Belgium, Canada, the Czech Republic, France, Germany, the United Kingdom, and the United States). Participants were generally healthy adult smokers (≥ 10 cigarettes/day) with ≥ 1 prior quit attempt (≥ 2 weeks) using varenicline and no quit attempts in ≤ 3 months; they were randomly assigned (1:1) to 12 weeks' varenicline (n = 251) or placebo (n = 247) treatment, with individual counseling, plus 40 weeks' nontreatment follow-up. The primary efficacy end point was the carbon monoxide-confirmed (≤ 10 ppm) continuous abstinence rate for weeks 9-12, which was 45.0% (varenicline; n = 249) vs. 11.8% (placebo; n = 245; odds ratio: 7.08; 95% confidence interval: 4.34, 11.55; P < 0.0001). Common varenicline group adverse events were nausea, abnormal dreams, and headache, with no reported suicidal behavior. Varenicline is efficacious and well tolerated in smokers who have previously taken it. Abstinence rates are comparable with rates reported for varenicline-naive smokers.
Trial registration: ClinicalTrials.gov NCT01244061.
Figures

References
-
- World Health Organization Tobacco. Fact Sheet no. 339 . < http://www.who.int/mediacentre/factsheets/fs339/en/ > ( 2013. Accessed 24 October 2013.
-
- Ockene J.K., et al. Relapse and maintenance issues for smoking cessation. Health Psychol. 2000;19:17–31. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

