HIV-1 Nef is transferred from expressing T cells to hepatocytic cells through conduits and enhances HCV replication
- PMID: 24911518
- PMCID: PMC4050050
- DOI: 10.1371/journal.pone.0099545
HIV-1 Nef is transferred from expressing T cells to hepatocytic cells through conduits and enhances HCV replication
Abstract
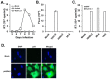
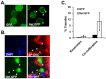
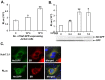
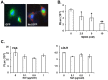
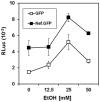
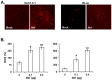
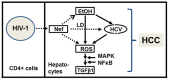
HIV-1 infection enhances HCV replication and as a consequence accelerates HCV-mediated hepatocellular carcinoma (HCC). However, the precise molecular mechanism by which this takes place is currently unknown. Our data showed that infectious HIV-1 failed to replicate in human hepatocytic cell lines. No discernible virus replication was observed, even when the cell lines transfected with HIV-1 proviral DNA were co-cultured with Jurkat T cells, indicating that the problem of liver deterioration in the co-infected patient is not due to the replication of HIV-1 in the hepatocytes of the HCV infected host. Instead, HIV-1 Nef protein was transferred from nef-expressing T cells to hepatocytic cells through conduits, wherein up to 16% (average 10%) of the cells harbored the transferred Nef, when the hepatocytic cells were co-cultured with nef-expressing Jurkat cells for 24 h. Further, Nef altered the size and numbers of lipid droplets (LD), and consistently up-regulated HCV replication by 1.5∼2.5 fold in the target subgenomic replicon cells, which is remarkable in relation to the initially indolent viral replication. Nef also dramatically augmented reactive oxygen species (ROS) production and enhanced ethanol-mediated up-regulation of HCV replication so as to accelerate HCC. Taken together, these data indicate that HIV-1 Nef is a critical element in accelerating progression of liver pathogenesis via enhancing HCV replication and coordinating modulation of key intra- and extra-cellular molecules for liver decay.
Conflict of interest statement
Figures

References
-
- Alter MJ (2006) Epidemiology of viral hepatitis and HIV co-infection. J Hepatol 44: S6–9. - PubMed
-
- Salmon-Ceron D, Lewden C, Morlat P, Bevilacqua S, Jougla E, et al. (2005) Liver disease as a major cause of death among HIV infected patients: role of hepatitis C and B viruses and alcohol. J Hepatol 42: 799–805. - PubMed
-
- Thein HH, Yi Q, Dore GJ, Krahn MD (2008) Natural history of hepatitis C virus infection in HIV-infected individuals and the impact of HIV in the era of highly active antiretroviral therapy: a meta-analysis. AIDS 22: 1979–1991. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

