Conformational analysis, molecular structure and solid state simulation of the antiviral drug acyclovir (zovirax) using density functional theory methods
- PMID: 24915059
- PMCID: PMC4078516
- DOI: 10.3390/ph7060695
Conformational analysis, molecular structure and solid state simulation of the antiviral drug acyclovir (zovirax) using density functional theory methods
Abstract
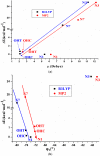
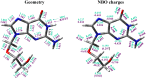
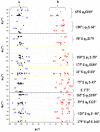
The five tautomers of the drug acyclovir (ACV) were determined and optimised at the MP2 and B3LYP quantum chemical levels of theory. The stability of the tautomers was correlated with different parameters. On the most stable tautomer N1 was carried out a comprehensive conformational analysis, and the whole conformational parameters (R, β, Φ, φ1, φ2, φ3, φ4, φ5) were studied as well as the NBO Natural atomic charges. The calculations were carried out with full relaxation of all geometrical parameters. The search located at least 78 stable structures within 8.5 kcal/mol electronic energy range of the global minimum, and classified in two groups according to the positive or negative value of the torsional angle j1. In the nitrogen atoms and in the O2' and O5' oxygen atoms of the most stable conformer appear a higher reactivity than in the natural nucleoside deoxyguanosine. The solid state was simulated through a dimer and tetramer forms and the structural parameters were compared with the X-ray crystal data available. Several general conclusions were emphasized.
Figures







References
-
- Schloemer G.C., Han Y.-K., Harrington P.J. Preparation of acyclovir using 1,3-dioxolane. 5567816. U.S. Patent. 1996 Oct 22;
-
- Lazrek H.B., Baddi L., Smietana M., Vasseur J.-J., Sebti S., Zahouily M. One-pot synthesis of antiviral acyclovir and other nucleosides derivatives using doped natural phosphate as Lewis acid catalyst. Nucleos. Nucleot. Nucleic Acids. 2008;27:1107–1112. doi: 10.1080/15257770802341285. - DOI - PubMed
-
- Gao H., Mitra A.K. Synthesis of acyclovir, ganciclovir and their prodrugs: A review. Synthesis. 2000;3:329–351.
-
- Emmert D.H., Lancaster M.D. Treatment of common cutaneous herpes simplex virus infections. Am. Fam. Physician. 2000;61:1697–1607. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

