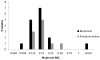Preventing surgical site infections: a randomized, open-label trial of nasal mupirocin ointment and nasal povidone-iodine solution
- PMID: 24915210
- PMCID: PMC4668802
- DOI: 10.1086/676872
Preventing surgical site infections: a randomized, open-label trial of nasal mupirocin ointment and nasal povidone-iodine solution
Abstract
Background: Treatment of Staphylococcus aureus colonization before surgery reduces risk of surgical site infection (SSI). The regimen of nasal mupirocin ointment and topical chlorhexidine gluconate is effective, but cost and patient compliance may be a barrier. Nasal povidone-iodine solution may provide an alternative to mupirocin.
Methods: We conducted an investigator-initiated, open-label, randomized trial comparing SSI after arthroplasty or spine fusion in patients receiving topical chlorhexidine wipes in combination with either twice daily application of nasal mupirocin ointment during the 5 days before surgery or 2 applications of povidone-iodine solution into each nostril within 2 hours of surgical incision. The primary study end point was deep SSI within the 3 months after surgery.
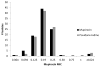
Results: In the modified intent-to-treat analysis, a deep SSI developed after 14 of 855 surgical procedures in the mupirocin group and 6 of 842 surgical procedures in the povidone-iodine group (P = .1); S. aureus deep SSI developed after 5 surgical procedures in the mupirocin group and 1 surgical procedure in the povidone-iodine group (P = .2). In the per protocol analysis, S. aureus deep SSI developed in 5 of 763 surgical procedures in the mupirocin group and 0 of 776 surgical procedures in the povidone-iodine group (P = .03).
Conclusions: Nasal povidone-iodine may be considered as an alternative to mupirocin in a multifaceted approach to reduce SSI.
Trial registration: ClinicalTrials.gov identifier: NCT01313182.
Conflict of interest statement
Conflict of Interest Disclosures
M. Phillips, 3M corporation (research grant which funded this study); had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
A. Rosenberg, 3M Corporation (research grant which funded this study)
B. Shopsin, 3M Corporation (research grant which funded this study) and grant funding is: R01 AI103268-01
G. Cuff, no potential conflicts of interest
F. Skeete, no potential conflicts of interest
A. Foti, no potential conflicts of interest
K. Kraemer, no potential conflicts of interest
K. Inglima, no potential conflicts of interest
B. Press, no potential conflicts of interest
J. Bosco, 3M Corporation (research grant which funded this study)
Figures
References
-
- Kurtz SM, et al. Infection risk for primary and revision instrumented lumbar spine fusion in the Medicare population. J Neurosurg Spine. 2012;17(4):342–7. - PubMed
-
- Kurtz SM, et al. Economic burden of periprosthetic joint infection in the United States. J Arthroplasty. 2012;27(8 Suppl):61–5 e1. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical