Structure-function of proteins interacting with the α1 pore-forming subunit of high-voltage-activated calcium channels
- PMID: 24917826
- PMCID: PMC4042065
- DOI: 10.3389/fphys.2014.00209
Structure-function of proteins interacting with the α1 pore-forming subunit of high-voltage-activated calcium channels
Abstract
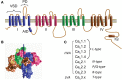
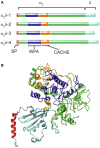
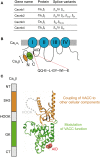
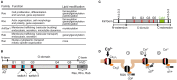
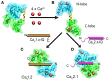
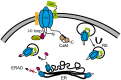
Openings of high-voltage-activated (HVA) calcium channels lead to a transient increase in calcium concentration that in turn activate a plethora of cellular functions, including muscle contraction, secretion and gene transcription. To coordinate all these responses calcium channels form supramolecular assemblies containing effectors and regulatory proteins that couple calcium influx to the downstream signal cascades and to feedback elements. According to the original biochemical characterization of skeletal muscle Dihydropyridine receptors, HVA calcium channels are multi-subunit protein complexes consisting of a pore-forming subunit (α1) associated with four additional polypeptide chains β, α2, δ, and γ, often referred to as accessory subunits. Twenty-five years after the first purification of a high-voltage calcium channel, the concept of a flexible stoichiometry to expand the repertoire of mechanisms that regulate calcium channel influx has emerged. Several other proteins have been identified that associate directly with the α1-subunit, including calmodulin and multiple members of the small and large GTPase family. Some of these proteins only interact with a subset of α1-subunits and during specific stages of biogenesis. More strikingly, most of the α1-subunit interacting proteins, such as the β-subunit and small GTPases, regulate both gating and trafficking through a variety of mechanisms. Modulation of channel activity covers almost all biophysical properties of the channel. Likewise, regulation of the number of channels in the plasma membrane is performed by altering the release of the α1-subunit from the endoplasmic reticulum, by reducing its degradation or enhancing its recycling back to the cell surface. In this review, we discuss the structural basis, interplay and functional role of selected proteins that interact with the central pore-forming subunit of HVA calcium channels.
Keywords: calcium channels; ion channels; protein complexes; subunit; voltage-dependent channels.
Figures

References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

