Does antigen masking by ubiquitin chains protect from the development of autoimmune diseases?
- PMID: 24917867
- PMCID: PMC4042494
- DOI: 10.3389/fimmu.2014.00262
Does antigen masking by ubiquitin chains protect from the development of autoimmune diseases?
Abstract
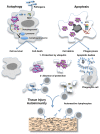
Autoimmune diseases are characterized by the production of antibodies against self-antigens and generally arise from a failure of central or peripheral tolerance. However, these diseases may develop when newly appearing antigens are not recognized as self by the immune system. The mechanism by which some antigens are "invisible" to the immune system is not completely understood. Apoptotic and complement system defects or autophagy imbalance can generate this antigenic autoreactivity. Under particular circumstances, cellular debris containing autoreactive antigens can be recognized by innate immune receptors or other sensors and can eventually lead to autoimmunity. Ubiquitination may be one of the mechanisms protecting autoreactive antigens from the immune system that, if disrupted, can lead to autoimmunity. Ubiquitination is an essential post-translational modification used by cells to target proteins for degradation or to regulate other intracellular processes. The level of ubiquitination is regulated during T cell tolerance and apoptosis and E3 ligases have emerged as a crucial signaling pathway for the regulation of T cell tolerance toward self-antigens. I propose here that an unrecognized role of ubiquitin and ubiquitin-like proteins could be to render intracellular or foreign antigens (present in cellular debris resulting from apoptosis, complement system, or autophagy defects) invisible to the immune system in order to prevent the development of autoimmunity.
Keywords: UBLs; apoptosis; autoimmunity; autophagy; immune tolerance; ubiquitin.
Figures
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

