Evolving use of new oral anticoagulants for treatment of venous thromboembolism
- PMID: 24923298
- PMCID: PMC4133479
- DOI: 10.1182/blood-2014-03-563056
Evolving use of new oral anticoagulants for treatment of venous thromboembolism
Abstract
The new oral anticoagulants (NOACs), which include dabigatran, rivaroxaban, apixaban, and edoxaban, are poised to replace warfarin for treatment of the majority of patients with venous thromboembolism (VTE). With a rapid onset of action and the capacity to be administered in fixed doses without routine coagulation monitoring, NOACs streamline VTE treatment. In phase 3 trials in patients with acute symptomatic VTE, NOACs have been shown to be noninferior to conventional anticoagulant therapy for prevention of recurrence and are associated with less bleeding. Rivaroxaban and dabigatran are already licensed for VTE treatment in the United States, and apixaban and edoxaban are under regulatory consideration for this indication. As the number of approved drugs increases, clinicians will need to choose the right anticoagulant for the right VTE patient. To help with this decision, this review (1) compares the pharmacologic profiles of the NOACs, (2) outlines the unique design features of the phase 3 trials that evaluated the NOACs for VTE treatment, (3) reviews the results of these trials highlighting similarities and differences in the findings, (4) provides perspective about which VTE patients should receive conventional treatment or are candidates for NOACs, and (5) offers suggestions about how to choose among the NOACs.
© 2014 by The American Society of Hematology.
Figures
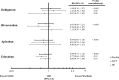
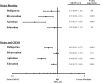
References
-
- White RH. The epidemiology of venous thromboembolism. Circulation. 2003;107(23 Suppl 1):I4–I8. - PubMed
-
- Silverstein MD, Heit JA, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ., III Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med. 1998;158(6):585–593. - PubMed
-
- Heit JA, Silverstein MD, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ., III Predictors of survival after deep vein thrombosis and pulmonary embolism: a population-based, cohort study. Arch Intern Med. 1999;159(5):445–453. - PubMed
-
- Kearon C. Natural history of venous thromboembolism. Circulation. 2003;107(23 Suppl 1):I22–I30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

