Age- and sex-dependence of dopamine release and capacity for recovery identified in the dorsal striatum of C57/Bl6J mice
- PMID: 24925086
- PMCID: PMC4055716
- DOI: 10.1371/journal.pone.0099592
Age- and sex-dependence of dopamine release and capacity for recovery identified in the dorsal striatum of C57/Bl6J mice
Abstract
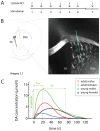
The dorsal striatum is the main input structure of the basal ganglia and the major target area of dopaminergic projections originating in the substantia nigra pars compacta. Heavily involved in the regulation of voluntary movement and habit formation, this structure is of strong importance in Parkinson's disease, obsessive-compulsive disorder, Tourette's syndrome and addiction. The C57/Bl6J mouse strain, the most commonly used strain in preclinical research today, is frequently used as a model organism for analysis of dopaminergic parameters implicated in human pathophysiology. Several components of the dopamine system have been shown to vary with age and sex, however knowledge of the contribution of these factors for dopamine release kinetics in the C57/Bl6J mouse strain is lacking. In the present study, we used an intracranial KCl-stimulation challenge paradigm to provoke release from dopaminergic terminals in the dorsal striatum of anaesthetized C57/Bl6J mice. By high-speed in vivo chronoamperometric recordings, we analyzed DA release parameters in male and female mice of two different ages. Our experiments demonstrate elevated DA amplitudes in adult compared to young mice of both sexes and higher DA amplitudes in females compared to males at both ages. Adult mice exhibited higher recovery capabilities after repeated stimulation than did young mice and also showed a lower variability in the kinetic parameters trise and t80 between stimulations. These results identified age- and sex- dimorphisms in DA release parameters and point to the importance of taking these dimorphisms into account when utilizing the C57/Bl6J mouse strain as model for neurological and neuropsychiatric disorders.
Conflict of interest statement
Figures


References
-
- Andén NE, Carlsson A, Dahlström A, Fuxe K, Hillarp NÅ, et al. (1964) Demonstration and mapping out of nigro-neostriatal dopamine neurons. Life Sciences 3: 523–530. - PubMed
-
- Obeso JA, Rodríguez-Oroz MC, Benitez-Temino B, Blesa FJ, Guridi J, et al. (2008) Functional organization of the basal ganglia: Therapeutic implications for Parkinson's disease. Movement Disorders 23: S548–S559. - PubMed
-
- Yin HH, Knowlton BJ (2006) The role of the basal ganglia in habit formation. Nature Reviews Neuroscience 7: 464–476. - PubMed
-
- Wightman RM, Robinson DL (2002) Transient changes in mesolimbic dopamine and their association with 'reward'. J Neurochem 82: 721–735. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

