Identification of a novel neurotrophic factor from primary retinal Müller cells using stable isotope labeling by amino acids in cell culture (SILAC)
- PMID: 24925906
- PMCID: PMC4159655
- DOI: 10.1074/mcp.M113.033613
Identification of a novel neurotrophic factor from primary retinal Müller cells using stable isotope labeling by amino acids in cell culture (SILAC)
Abstract
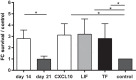
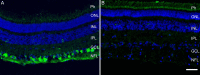
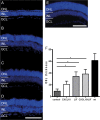
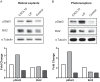
Retinal Müller glial cells (RMGs) have a primary role in maintaining the homeostasis of the retina. In pathological situations, RMGs execute protective and regenerative effects, but they can also contribute to neurodegeneration. It has recently been recognized that cultured primary RMGs secrete pro-survival factors for retinal neurons for up to 2 weeks in culture, but this ability is lost when RMGs are cultivated for longer durations. In our study, we investigated RMG supernatants for novel neuroprotective factors using a quantitative proteomic approach. Stable isotope labeling by amino acids in cell culture (SILAC) was used on primary porcine RMGs. Supernatants of RMGs cultivated for 2 weeks were compared with supernatants from cells that had already lost their protective capacity. Using this approach, we detected established neurotrophic factors such as transferrin, osteopontin, and leukemia inhibitory factor and identified C-X-C motif chemokine 10 (CXCL10) as a novel candidate neuroprotective factor. All factors prolonged photoreceptor survival in vitro. Ex vivo treatment of retinal explants with leukemia inhibitory factor or CXCL10 demonstrated a neuroprotective effect on photoreceptors. Western blots on CXCL10- and leukemia inhibitory factor-stimulated explanted retina and photoreceptor lysates indicated activation of pro-survival signal transducer and activator of transcription signaling and B-cell lymphoma pathways. These findings suggest that CXCL10 contributes to the supportive potential of RMGs toward retinal neurons.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

References
-
- Schnitzer J. (1988) Astrocytes in the guinea pig, horse, and monkey retina: their occurrence coincides with the presence of blood vessels. Glia 1, 74–89 - PubMed
-
- Ashwell K. W., Hollander H., Streit W., Stone J. (1989) The appearance and distribution of microglia in the developing retina of the rat. Vis. Neurosci. 2, 437–448 - PubMed
-
- Bringmann A., Pannicke T., Grosche J., Francke M., Wiedemann P., Skatchkov S. N., Osborne N. N., Reichenbach A. (2006) Muller cells in the healthy and diseased retina. Prog. Retin. Eye Res. 25, 397–424 - PubMed
-
- Reichenbach A., Bringmann A. (2013) New functions of Muller cells. Glia 61, 651–678 - PubMed
-
- Willbold E., Berger J., Reinicke M., Wolburg H. (1997) On the role of Muller glia cells in histogenesis: only retinal spheroids, but not tectal, telencephalic and cerebellar spheroids develop histotypical patterns. J. Hirnforsch 38, 383–396 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

